The reduction in risk of cardiac death offered by revascularisation in patients with moderate to large amount of stress-induced myocardial ischaemia has driven advancements in percutaneous coronary intervention (PCI) technology over the last four decades.1 However, despite significant progress in the techniques, equipment and pharmacotherapy, target lesion failure remains the Achilles heel of a PCI approach in patients with coronary heart disease.
The advent of the bare metal stent (BMS) introduced a major shift and promised improved outcomes over percutaneous balloon angioplasty (POBA). The BMS prevented the elastic recoil and constrictive remodelling that was seen frequently with POBA (32–55% incidence).2,3 However, it was soon realised that the benefits of deploying a metallic scaffold were still accompanied with a significant (17–41%) incidence of restenosis within the stented segment.4–7
Further research and development in stent technology led to the emergence of drug-eluting stents (DES), with successive generations produced on platforms with different anti-proliferative drugs, advanced polymers, improved stent cell design and thinner metallic struts. This promised to solve the spectre of in-stent restenosis (ISR) completely by preventing early tissue formation after stent deployment. These improvements have certainly led to superior results with reduced target lesion failure and target lesion revascularisation, MI and stent thrombosis when compared with BMS or the earlier generation of DES.8,9
However, despite these major developments, the incidence of DES ISR remains between 5 and 10% and is an independent predictor of mortality, thereby making it the foremost adversary of an interventional cardiologist in the modern era.10,11 This review highlights a simplified approach for identifying the mechanism of ISR and describe strategies to select devices for therapy and illustrate this with clinical cases (Figures 1–6).
Definition of In-stent Restenosis
ISR is angiographically defined as >50% reduction in luminal area within the stent or in the adjacent native vessel (5 mm of the proximal or distal stent edge).12 The clinical definition, however, includes the angiographic appearance and the presence of one of the following:
- clinical symptoms suggestive of coronary heart disease;
- ECG changes suggestive of underlying coronary ischaemia;
- significant limitation in coronary flow as measured by a positive haemodynamic assessment such as fractional flow reserve or instantaneous wave-free ratio (iFR);
- minimum cross-sectional area of <4 mm2 (6 mm2 for left main stem) using intravascular ultrasound; or
- a reduction of >70% in luminal area, even in the absence of symptoms.13
Mehran’s classification system was developed for morphological classification of BMS ISR, but it has also shown prognostic value in DES ISR as well.14,15 As per the classification, the ISR is described to be focal, diffuse, proliferative or occlusive, and it helps in predicting the rate of revascularisation (19%, 35%, 50% and 98%, respectively).14
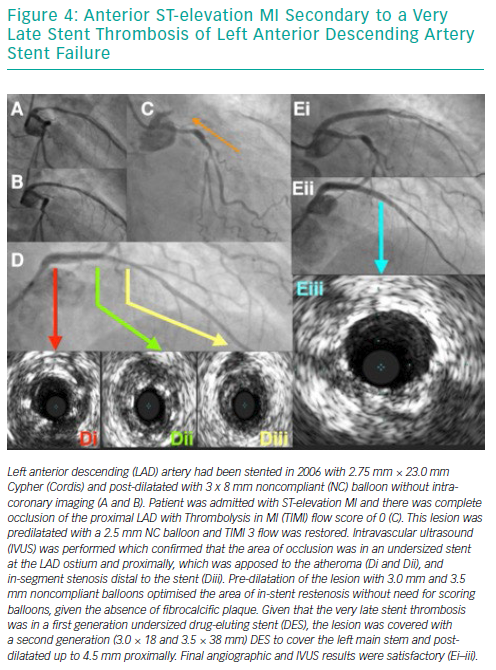
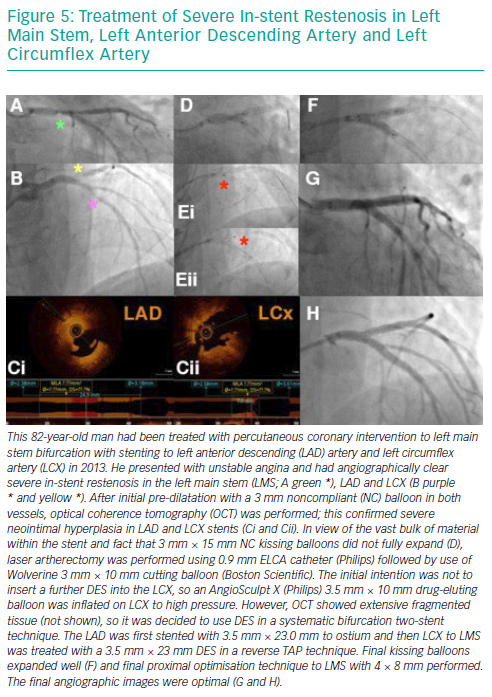
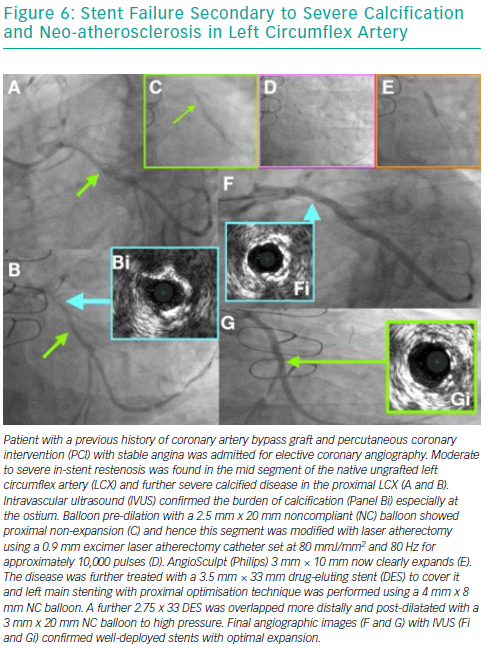
Risk Factors for Developing In-stent Restenosis
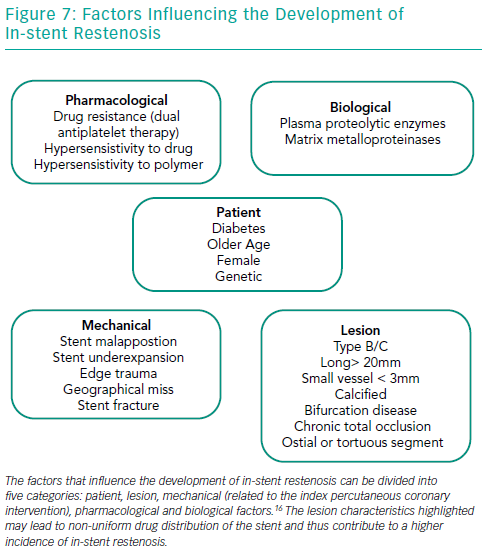
Several factors play important roles in the development of ISR in BMS and DES (Figure 7). Diabetes is perhaps the most well-established patient risk factor for ISR, particularly with BMS – the rate of BMS ISR may be as high as 30–50%.16–19 There are various lesion characteristics that lead to non-uniform drug distribution and thus contribute to a higher incidence of ISR.
The presence of moderate or severe calcification is perhaps one of the most challenging aspects of PCI in contemporary practice. There is clear evidence that the degree of lesion calcification directly affects stent expansion. In many large-scale clinical studies, calcification has been shown to be proportionally linked to stent failure, with increased rates of target lesion failure, target vessel revascularisation, MI and death in patients with the most lesion calcification.20,21 Advancing a stent through a calcified tortuous vessel may lead to disruption of polymer and/or drug on the surface, which can reduce the efficacy of even the best-designed DES.
PCI of long lesions (>20 mm) and small calibre vessels (<3 mm and especially those <2.5 mm) carries a much higher risk of ISR and such characteristics are often seen when treating chronic total occlusion. The risk of ISR doubles if the length of the stented segment is >35 mm compared to <20 mm.12,22,23 The relation of vessel diameter to ISR was reported in the Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction (HORIZONS-AMI) trial, where vessel size <3 mm was related to a significantly higher incidence of ISR.24 Bifurcation lesions, especially those treated with a double stent (the main vessel and side branch technique), have a higher incidence of stent failure, particularly in the side branch.25
Pathophysiology of In-stent Restenosis
It has been observed that ISR secondary to BMS versus DES has different characteristics, with important ones being time lag from stent implantation to presentation, morphology of the ISR itself and response to treatment.26,27 BMS ISR presents early (typically 6–8 months) as compared to DES ISR (typically after 2 years) which often has a delayed presentation.28
The initial inflammatory process ensues soon after the stent is implanted, and is characterised by deposition of platelets and fibrin, as well as adhesions of circulating neutrophils and macrophages. Over several weeks these cells are replaced by chronic inflammatory cells, which include macrophages and giant cells. Simultaneously, this vascular injury from the stent struts in the intima induces the initial stimuli for vascular smooth muscle cell proliferation and activation. As a result, the vascular smooth muscle cells migrate from the tunica media, and the myofibroblasts migrate from the tunica adventitia into the tunica intima, forming an extracellular matrix. This is proven by the systemic surge in the levels of the inflammatory markers post PCI and also by the presence of inflammatory cells in the plaque.29 These processes culminate in the formation of a neointimal layer over the stented segment, with its luminal side covered by the endothelial cells.22,30
DES ISR is characterised by delayed healing of the vessel wall secondary to stent components such as the durable polymer. Though the durable polymer facilitates drug delivery, it also results in a chronic non-specific inflammatory process (especially the durable polymer on first generation DES), which results in incomplete neo-endothelialisation, and occasionally can cause a specific hypersensitivity reaction.31 This led to the development of biodegradable polymers, but recent data have suggested similar safety and efficacy of biodegradable polymer DES compared to second generation durable polymer DES.32
The above pathogenic processes lead to different time of onset and morphological characteristics. While BMS ISR peaks around 3–6 months after stent implantation and has a diffuse pattern of neointima formation, DES ISR has a predominantly focal pattern, with onset after 6–9 months and increasing up to 2 years after implantation.31,33
Neo-atherosclerosis
When describing the pathophysiology of ISR, it is important to understand the process of neo-atherosclerosis. As with native vessel, the atherosclerotic process can affect neointima as well. This occurs due to incomplete endothelialisation, which is seen more commonly in DES as compared to BMS, primarily due to the elution of the drug itself.34,35 This results in uptake of circulating lipids and formation of plaque, which is thin-capped and occurs earlier in DES than BMS (2 years versus 6 years, respectively).34 There are several independent risk factors that lead to neo-atherosclerosis: young age, longer duration after stent implantation, sirolimus or paclitaxel-eluting stents, smoking, chronic kidney disease and LDL-cholesterol >3.9 mmol/l.34
ISR was earlier considered to be a benign clinical pathology, but can can present as acute coronary syndrome (ACS).36,37 Magalhaes et al. found that the incidence of ACS in the patient presenting with DES-ISR (second-generation DES) requiring target vessel revasculariation was 66.7%, and MI was 5.2%.38 This occurs as a result of an acceleration of the neo-atherosclerotic process, which culminates in plaque rupture and thrombus formation, possibly manifesting as late stent thrombosis.39 It is also important to remember that stable patients with ISR have a favourable prognosis, and should be assessed with contemporary validated technologies such as pressure wire before undertaking PCI.40,41
Diagnosis and Evaluation of In-stent Restenosis
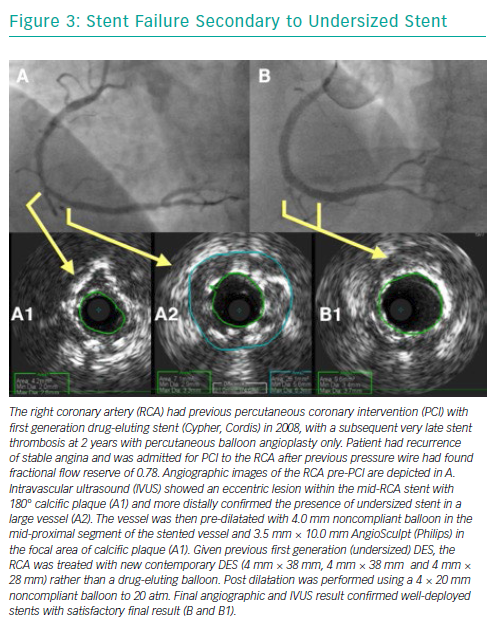
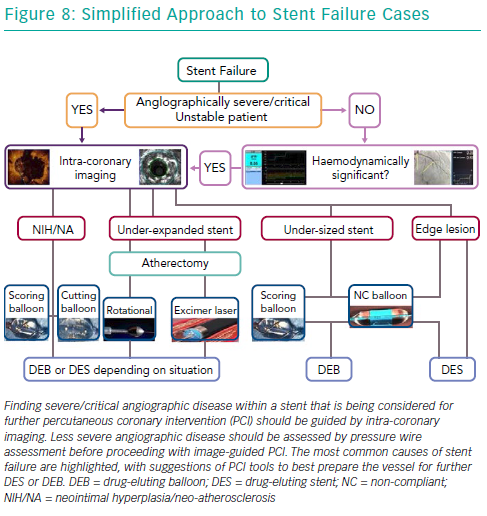
Selective coronary angiography is the initial diagnostic tool to diagnose and assess ISR, despite its limited resolution. Although modern features of fluoroscopic equipment, such as stent enhancement, permit diagnosis of an underexpanded stent, it is rare for coronary angiography alone to provide sufficient insight into the mechanism of stent failure. Intra-coronary imaging tools such as intravascular ultrasound and optical coherence tomography (OCT) are now recommended for PCI for stent failure, since either imaging technique allows detailed assessment of the native vessel and stented segment to provide precise mechanistic information (Figure 8).42 Such factors that might easily be identified are stent undersizing, underdeployment or underexpansion, geographical miss of the lesion and stent fracture.43,44 Intra-coronary imaging also assists the visualisation of neo-intimal hyperplasia, neo-atherosclerosis, edge stenosis, underlying calcification and provides clear instruction on what devices are necessary to prepare the lesion and then accurately size and expand the stent.45 Evidence supports this approach. For example, intravascular ultrasound-guided revascularisation has been shown to provide better clinical and angiographic results,46,47 with a 1 mm2 increase in minimal stent area found to be associated with a 20% decrease in BMS ISR.27,48
OCT has a better axial resolution (15 μm), which helps to morphologically differentiate between the homogenous high signal tissue band of BMS (constituted by neointimal hyperplasia which is rich in vascular smooth muscle cells) and the heterogeneous, focal and layered tissue band of DES (rich in proteoglycan and fibrin content).27,49
Also, before considering therapy on angiographic diagnosed ISR in stable patients, it is important to assess whether the lesion is causing ischaemia and guide therapy using adjunctive and validated technology such as pressure wire (Figure 8).40,41 It has been previously shown that coronary angiography alone correlates poorly with the functional significance of moderate ISR lesions.41,50 With the advent of the iFR and SyncVision technology, it is now possible to simultaneously assess the functional significance of the lesion, measure the length of the expected stented segment and predict the post revascularistion iFR, all of which can be performed without inducing hyperaemia.51
Treatment of In-stent Restenosis
Bare Metal Stent In-stent Restenosis
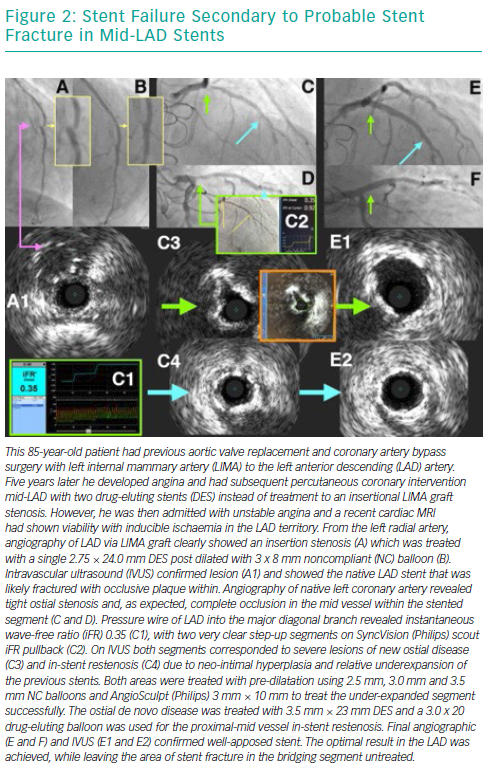
Over the years, several advancements have been made in the treatment of ISR with an initial focus on BMS-ISR, which had a high incidence rate.4–6 Identification of the mechanism of ISR is critical to the understanding of how best to deal with the lesion. For instance, an undersized stent with minimal intra-luminal material may best be optimised by just balloon dilatation (Figure 8). More complex mechanisms of ISR such as severe neointimal hyperplasia or neo-atherosclerosis may require debulking strategies, using tools such as scoring balloons or atherectomy (Figure 2). There have been many studies comparing alterative PCI strategies for treatment of ISR (Table 1).
Two trials studying the role of rotational atherectomy in treatment of BMS ISR produced conflicting results. Rotational atherectomy had significantly lower target lesion failure rates in the Rotational Atherectomy Versus Balloon Angioplasty for Diffuse In-stent Restenosis (ROSTER) trial, while POBA had significantly lower restenosis in the Angioplasty Versus Rotational Atherectomy for Treatment of Diffuse In-stent Restenosis Trial (ARTIST).52,53
The use of excimer laser atherectomy confers several advantages, such as the ability to modify plaque behind stent struts, decreased potential risk of distal emboli and lower risk of stent fracture or entrapment.54–56 These advantages have translated into superior outcomes such as greater acute luminal gain when treating complex DES ISR, as recently reported by Ichimoto et al.57 In chronically occluded ISR or where there is an inability to cross the lesion with disease-modifying devices, excimer laser atherectomy is the better option.
Once the existing stent has been adequately optimised, the next decision is how to prevent future ISR due to vessel injury and provide a long-term durable solution. The use of a drug-eluting balloon (DEB) potentially confers certain advantages over a DES. These include homogenous distribution of the drug in the vessel wall (especially if the original stent was suboptimally expanded), absence of polymer leading to reduction in the chronic inflammatory process, and reduced number of layers of the stent struts.58 The clinical and angiographic advantage of paclitaxel-eluting balloon (PEB) compared with POBA and PES in the treatment of BMS ISR was shown in the Treatment of In-stent Restenosis by Paclitaxel Coated PTCA Balloons (PACCOCATH ISR) I and II and Paclitaxel-Eluting PTCA-balloon catheter in Coronary Artery Disease (PEPCAD) II trials, respectively.59–61 The role of PEB in treatment of BMS ISR was further established when it demonstrated comparable results against the everolimus-eluting stent (EES) in the Restenosis Intra-Stent of Bare Metal Stents (RIBS) V and Treatment of In-Stent restenosis (TIS) trials.62,63
The use of DES in the treatment of BMS ISR was evaluated and firmly confirmed by the Sirolimus-Eluting Stent for In-Stent Restenosis (SISR) and the TAXUS Paclitaxel-Eluting Coronary Stent in the Treatment of In-Stent Restenosis (TAXUS V ISR) trials, both revealing lower rates of binary restenosis and better clinical outcomes with DES compared to complex brachytherapy.64,65 Similarly, when DES was compared to POBA for treating BMS ISR, it showed superior results in the ISAR-DESIRE and RIBS II trial.66,67
Drug-eluting Stent In-stent Restenosis
DES ISR is associated with worse outcomes than BMS ISR, and this has led to the development of different treatment strategies using DES or PEB.68,69 Lesion preparation in the treatment of -limus DES ISR was studied in the Intracoronary Stenting and Angiographic Results: Drug-Eluting Stents for In-Stent Restenosis (ISAR-DESIRE) 4 trial, where the use of a scoring balloon before DEB resulted in a significantly lower percentage of diameter stenosis and restenosis rate compared to POBA.70 This difference is contributed by better precision, power (15–25 times higher than POBA), uniform expansion and safety (lower dissection and perforation rates) of the angiosculpt scoring balloon compared to POBA.71–73
Given that most contemporary cases of ISR are in DES and not BMS, the option of simply re-treating the lesion with another DES is usually not ideal. As described above, DEB offers several advantages, and these have been established in the treatment of DES ISR as well. PEB was found to be better or equally effective in treating DES ISR when compared to POBA or PES, as studied in the PEPCAD-DES and PEPCAD China ISR and ISAR-DESIRE 3 trials, respectively.74–76 Similarly, Naganuma et al. reported no difference in the target vessel revascularisation and MACE endpoints, when bifurcation BMS/DES ISR was treated using either EES or PEB.77 When PEB was compared to EES in the treatment of DES ISR, conflicting results were revealed by the Drug-Eluting Balloon for In-Stent Restenosis (DARE) trial and the recently published 3-year outcome data from the RIBS IV trial.78,79 Thus there is a sufficient body of evidence supporting the use of DEB in the treatment of DES ISR where clinically suited and indicated.
Treating DES ISR secondary to stent undersizing, edge dissection or stent fracture is best treated using another DES. The role of similar DES (homo) or different DES (hetero) has been evaluated to understand if a similar or different anti-proliferative drug offers any advantage. This has been studied in the ISAR-DESIRE 2, New Generation Drug Eluting Stent for In-stent Restenosis of Drug Eluting Stent (RESTENT-ISR) and RIBS III trials.80–82 While the ISAR-DESIRE 2 and RESTENT-ISR revealed no significant difference between the use of homo or hetero stents, RIBS III found significantly better clinical and angiographic outcomes in the hetero-DES group.
An alternative concept to the repeated use of DES when a DEB alone is considered inadequate has been to consider bioresorbable devices. This could potentially offer the opportunity of treating ISR without implanting long-term multiple layers of stents (known as the ‘onion skin’). Absorb (Abbott Vascular) had been the most widely used bioresorbable vascular scaffold since first-in-man studies in simple de novo lesions in 2006.83
In recently published literature, rates of target lesion failure rates at 12 months of 9.1–12.2% have been reported with bioresorbable vascular scaffolds in the treatment of BMS/DES ISR.84,85 Although used by some operators in ISR cases, the relative large strut thickness (160 μm), footprint and need for near-perfect lesion preparation significantly restricted use in stent failure for the majority of BVS implanters. Absorb was removed from the market in 2017 after several studies pointed to increased scaffold thrombosis rates compared to DES and failure to match target lesion failure/target vessel revascularisation rates within the first 3 years while the device resorbed.
Conclusion
Stent failure through in-stent restenosis remains an occurrence that interventional cardiologists will face on a routine basis. Utilisation of diagnostic tools, such as pressure wire assessment and intracoronary imaging, provide better insights compared with angiography alone, and permit more focussed therapies to treat these lesions. The repeat revascularisation often requires adjunctive devices to optimise the outcome and provide long-term durable result. Although data are available to currently support the PCI strategies that we have discussed in this paper, further research will be necessary to distinguish which are the superior PCI techniques within this heterogeneous patient cohort.