Dr Kanwar’s talk was focused on sharing a real-world clinical experience in heart failure recovery with temporary and durable left ventricular assist devices (LVAD). Dr Kanwar noted that until recently, discussions and terminology on LVAD, heart failure and cardiogenic shock were siloed and considered separately, rather than noting and exploring the connections between these conditions and therapies.
Dr Kanwar explained the notion of ‘recovery disconnect’, where carefully curated patient selections can result in 50% survival in studies such as RESTAGE-HF,1 but in large-scale databases, such as INTERMACS, survival to explant may be as low as 1–2%.2
Furthermore, although there are more patients recommended for and receiving heart transplants, there are fewer patients being removed from the transplant list or reported as on a path to recovery. This suggests that patients may be ‘rushed’ to transplant without enough time with an LVAD to determine the likelihood of recovery, especially since the United Network for Organ Sharing allocation policy change.3
As an example, Dr Kanwar described a case of a patient who, upon review, most clinicians recommended for urgent transplant; however, Dr Kanwar and her team instead proceeded with a durable LVAD as a bridge to transplant and began guideline-directed medical therapy (GDMT). The device was explanted 11 months later, and the patient’s left ventricular ejection fraction remains at 55% 3 years later.
This concept for providing ample time and support for recovery is not new. The use of mechanical circulatory support (MCS) as a bridge to recovery may promote myocardial recovery, resulting in higher rates of clinically successful device explants. As such, LVAD optimisation must be personalised, with the goal of recovery rather than transplant. Patients should be evaluated on a case-by-case basis to determine the haemodynamic needs and reversibility of the condition, and then therapies, particularly unloading devices, must be tailored to the specific needs of the patient. LVAD can be used as a durable support and bridge to recovery, as well as a bridge to transplant.
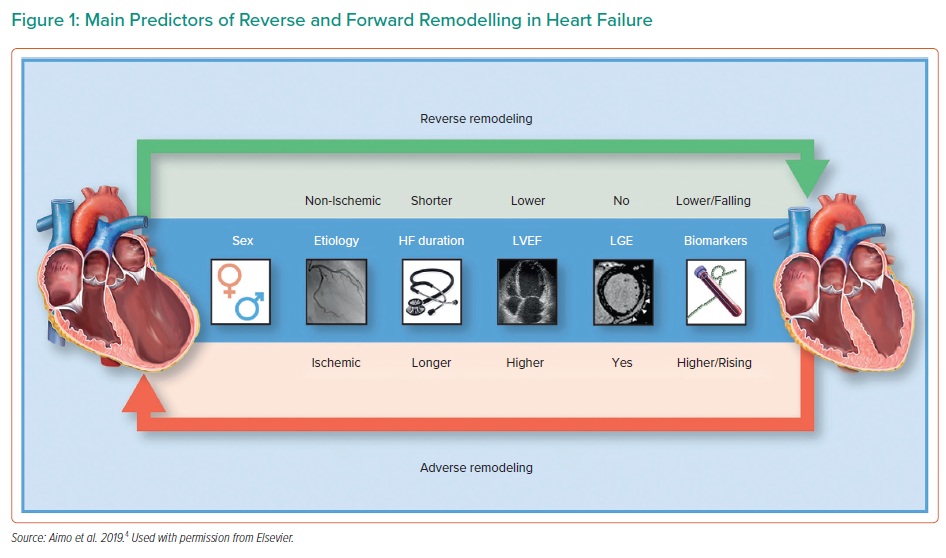
For the various types of MCS, there are different considerations depending on the goals of the therapy. Patients must be carefully phenotyped with regard to their likelihood of recovery (Figure 1); their long-term outcome goals need to be considered to determine the therapeutic aims. Temporary MCS provides early and upfront unloading, rhythm control, non-cardiac end-organ management, patient mobility and improved quality of life. Furthermore, it allows for time to monitor the patient’s metrics and determine an exit strategy.
Durable LVAD provides a long timeline, where appropriate patients are optimised and prepared carefully before, during and after device implantation. The patients should receive appropriate GDMT with careful monitoring, and device explantation should be considered upon long-term improvement.
Dr Kanwar concluded that biology is not binary and recovery is not just about device explantation. Important consideration must be given to the specifics of each patient in order to determine the best path for their specific medical needs and goals, with transplant as a last option rather than a primary goal.