Dr Wilcox’s talk focused on the differences between myocardial recovery and remission, and how to differentiate and target patient treatment between the two. Historically, heart failure (HF) has been considered as a steady track towards mortality, but the evidence from the field clearly demonstrates that HF is not a death sentence, and that recovery and remission are possible. HF is a syndrome of repeated hospital admissions, with failure to handle salt and water normally and accompanying neurohormonal abnormality. Given these components, the goal for treating HF patients should be to improve ejection fraction and to aid recovery from cardiomyopathy.
Traditionally, guideline-directed medical therapy (GDMT) for HF includes neurohormonal therapy, which reduces HF symptoms, reverses cardiac remodelling and improves survival. However, because the definition of cardiomyopathy recovery is constantly changing, it can be easier to define what HF with recovered ejection fraction (HFrecEF) is not, which is constantly evolving with better information and understanding of the HF recovery process. For example, HF with a midrange ejection fraction (45%) is not considered recovery, but rather is a biology that is associated with reverse remodelling and a reduction in left ventricular volume.1
Dr Wilcox presented a working definition of HFrecEF, where recovery is an improvement in left ventricular ejection fraction (LVEF) to an absolute value >40%, with at least a >10% improvement from the patient’s baseline, accompanied by reductions in ventricular volumes. The primary difference between remission and recovery is in the myocardial substrate, with persistent abnormalities in remission but ‘normal’ structure and function in recovery.2 Essentially, until the myocardium has become completely non-viable (i.e. late gadolinium scar, dead muscle), that myocardium should be a target for therapy and recovery.3
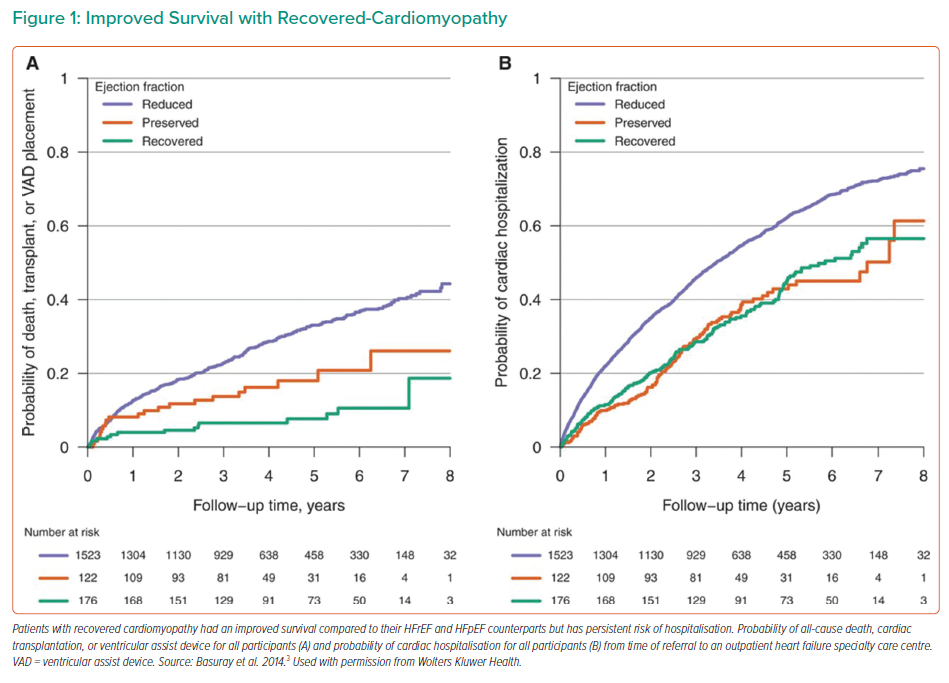
Dr Wilcox noted that hearts that have recovered from HF are still vulnerable to relapse; this is largely due to the fact that during recovery gene transcription becomes normalised, but the networks that had been persistently dysregulated will assume a novel biological set-point. As such, although recovery is associated with improved survival (Figure 1A), a persistent risk of hospitalisation still exists (Figure 1B).4 Dr Wilcox also noted that there are a number of factors to predict whether a patient is likely to recover from HF, including clinical parameters, genetic factors, echocardiographic/cardiac MRI and biomarker profile.5 In particular, non-ischaemic aetiology, shorter duration of HF and no left bundle branch block are indicators of reverse left ventricular (LV) remodelling with GDMT. In addition, well-preserved global longitudinal strain, a measure of LV systolic function that has been shown to be more reproducible than LVEF, can indicate that even a dilated patient may be able to achieve recovery, whereas abnormal global longitudinal strain can indicate future deterioration. Even among patients who achieve the traditional >40% LVEF, more normal global longitudinal strains are still indicative of long-term success, and even small incremental improvements can have long-term effects.5
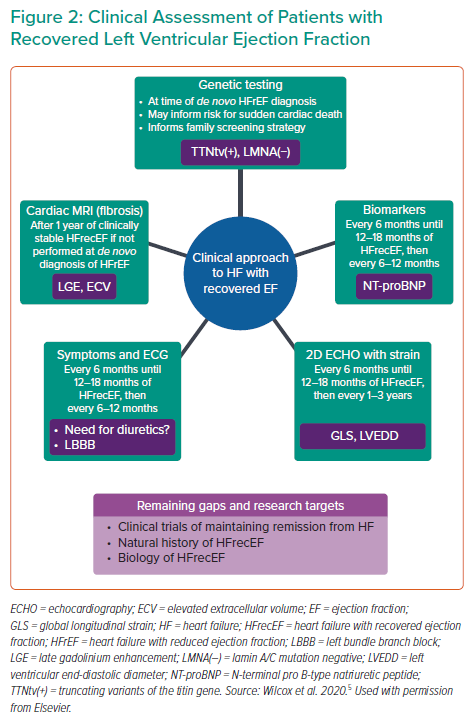
Patients who have recovered from HF still require maintenance in the form of medical therapies. The TRED-HF study examined withdrawal of pharmacological treatment for HF in patients with recovered dilated cardiomyopathy and revealed that cessation of medication resulted in a 44% rate of recrudescent HF within 8 weeks, requiring the resumption of medications. Thus, there are numerous angles from which to clinically approach HFrecEF, to understand personalised risks and improve long-term survival (Figure 3).5
Dr Wilcox proposed that recovery can, and should, be a medical target for HF patients. Impella 5.5 can be a helpful tool in achieving this, and is effective across many relevant indications, including cardiomyopathy cardiogenic shock, acute MI with cardiogenic shock and post-cardiotomy cardiogenic shock. Improvements with Impella 5.5 are robust and most patients can achieve improvement to allow for device removal. Future goals include novel devices that are specifically targeted to recovery, such as the developing Impella BTR (bridge-to-recovery), which received conditional investigational device exemption approval in January 2022 for a first-in-human early feasibility study.
Dr Wilcox concluded that although the goal should be to achieve recovery, achieving remission is an excellent result, and using temporary unloading devices as a platform for neurohormonal therapy may be the future for HF recovery and remission.