The Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) tracks the outcomes of all implantable devices in the US. According to this registry, the rates of device explantation, a measure of native heart recovery, is extremely low in patients with a left ventricular assist device (LVAD). Dr Birks noted that this is due, in part, to the fact that long-term devices are typically inserted as a bridge-to-transplant strategy. Furthermore, treating centres often lack testing for underlying myocardial function, pump speed optimisation to maximise unloading and drug therapy to promote recovery.
Conversely, when clinicians are using LVADs to attempt to reverse chronic heart failure and induce myocardial recovery, there are numerous crucial steps to take, including: regular, safe, and reliable testing; pump speed increases and drug additions to shrink the heart; medications and techniques designed to maximise recovery durability; and novel explant techniques engineered to minimise trauma to the heart.
In particular, targeted adjuvant therapies that enhance LVAD reverse remodelling are crucial for recovery. The aims of these therapies are to shrink the heart and induce reverse remodelling, enhance contractility, enhance recovery durability and counteract fibrosis. Dr Birks emphasised that she has found aggressive drug therapy in tandem with LVAD support to be critical for recovery, along with regular testing both before and after device removal.1 Over the years and across studies, device explantation rates have been low (3–24%), but most of these studies included very little or no HF medication therapy and none were optimised as part of the protocol.2–6
Conversely, studies such as Dr Birks’ that have included aggressive medical therapy in addition to LVAD reveal vastly increased rates of explanted devices of between 50% and 73%.7,8 More recent studies have provided further evidence that aggressive medical therapy improves survival and quality of life, regardless of patient or pump, suggesting that medical therapy is important in the population regardless of therapeutic aims.9
The Remission from Stage D Heart Failure (RESTAGE-HF) study sought to determine whether the aggressive attempts to maximise reverse remodelling and recovery with optimised LVAD and drug therapy are consistently reproducible. This uniform, prospective analysis was performed across six centres with existing experience of recovering patients, and with specific interest in recovery. The primary objective was to determine the proportion of patients who had sufficient improvement in ventricular function after standardised LVAD and pharmacological therapy and testing to allow removal of the LVAD within 18 months. Furthermore, patients were followed for up to 3 years to determine the durability of sustained remission, ultimately to identify predictors of recovery.10
Dr Birks noted that the 18-month window was not necessary, because most recruited patients were on destination therapy, and it is likely that future studies would not include this limit. The device explant criteria at zero flow (6,000 RPM) were: left ventricular (LV) end-diastolic diameter (LVEDD) <60 mm, LV end-systolic diameter <50 mm, LV ejection fraction >45%; LV end-diastolic pressure or pulmonary capillary wedge pressure ≤15 mmHg; resting cardiac index >2.4 l/min/m2; and with or without myocardial volume oxygen >16 ml/kg/min. Dr Birks and her team hypothesised that >10% of patients would achieve the primary endpoint of being free from mechanical circulatory support or heart transplant by 1 year after LVAD removal.
Dr Birks described how she and her team used echocardiography early and often in the patient care timeline to optimise the LVAD pump speed for each patient and to monitor LV function. Pump speed was increased in increments of 200 RPM while maintaining blood pressure <100 mmHg. This was continued until LVEDD was <60 mm and mitral regurgitation was Grade 2 or less.
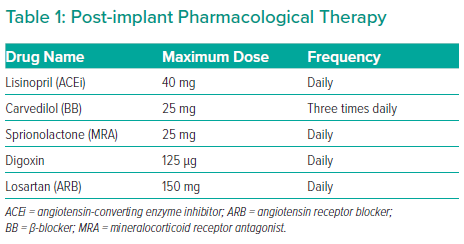
In addition, a combination of drugs was used at varying frequencies, but notably not clenbuterol (Table 1). Once the echocardiography explant criteria were met, patients underwent right heart catheterisation to test how well the heart was pumping, on normal and low flow, for 15 minutes.
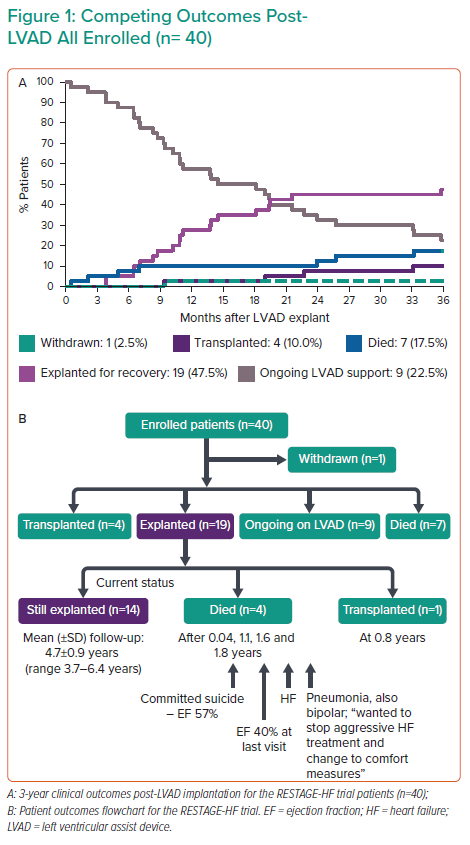
Of 40 patients, 19 were ultimately explanted for recovery (47.5%; Figure 1A). Furthermore, of the 19 explanted patients, 17 were still in remission after >1 year, one had died from unrelated causes and one was transplanted. Overall, 16 of 40 patients (40%) achieved the primary endpoint, far and above the 10% needed to reject the null hypothesis, and long-term follow-up revealed that 14 of 40 patients were still explanted at a mean (± SD) of 4.7 ± 0.9 years, demonstrating robust long-term recovery (Figure 1B).
Dr Birks expanded on the study outcomes, with univariate and multivariate analyses revealing that a predischarge LVEDD of <60 mm and a reduction in LVEDD of >7.5 mm between pump implantation and early after explantation were associated with a higher likelihood of having the device explanted and achieving recovery. Together, these findings suggest that those patients who respond early after device implantation with shrinking heart size have the best chance of ultimately recovering.
Dr Birks summarised that it is possible for patients in late-stage chronic heart failure to progress to recovery and eventual LVAD removal with aggressive medical and LVAD therapy. She concluded by emphasising that there is more to be done to optimise pumps and their use, and to identify novel medical approaches to enhance reverse remodelling and improve long-term patient outcomes and quality of life.