Anomalous aortic origin of a coronary artery (AAOCA) is a rare congenital anomaly and potential cause of sudden cardiac death.1 Patients with an interatrial course have traditionally been stratified as those at highest risk. However, there are many factors that participate in limiting coronary blood flow.2–4 The standard of care for symptomatic patients is surgical management with coronary unroofing.5 However, not all cases are amenable to surgery.
Case Report
A 69-year-old man with non-ischaemic cardiomyopathy with recovered left ventricular ejection fraction and an ICD presented after feeling his ICD fire. Relevant medical problems included paroxysmal AF, severe chronic obstructive pulmonary disease and a history of stage 1 adenocarcinoma of the lung. The patient was reportedly ambulating from the bathroom without wearing his home oxygen when he experienced a “jolt” in his abdomen associated with nausea/vomiting and dyspnoea. On initial evaluation, the patient was mildly hypotensive (blood pressure 102/58 mmHg) with an irregular heart rate of 79 BPM. Oxygen saturation by pulse oximetry was 92% on 3 l of oxygen by nasal cannula, and auscultation of his lungs revealed diffuse wheezing. The left subclavian ICD pocket was intact, and he was noted to have 1+ oedema of the lower extremities. The remainder of his physical examination was unremarkable.
The patient’s medical history included non-ischaemic cardiomyopathy stage C with New York Heart Association functional class II symptoms, ICD implantation for primary prevention (on goal-directed medical therapy, furosemide and digoxin), paroxysmal AF, hypertension and diabetes. Additional medical problems included severe emphysematous disease, as well as stage 1a adenocarcinoma of the lung treated by stereotactic body radiation and he was recently started on home oxygen therapy.
Differential diagnosis on presentation included hypoxia-induced arrhythmia, acute coronary syndrome, acute on chronic heart failure exacerbation, digoxin toxicity and pulmonary embolism.
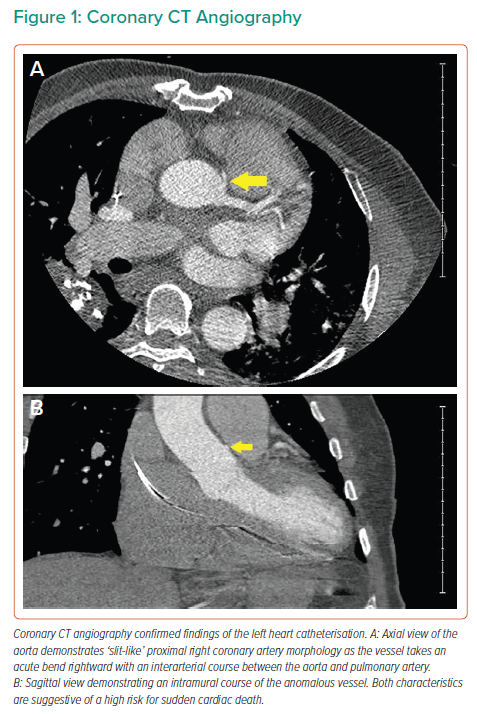
Initial laboratory evaluation, including serial troponins and electrolytes, was unrevealing. The digoxin level was therapeutic and electrocardiogram demonstrated rate-controlled AF. ICD interrogation revealed several episodes of VF, one terminated by antitachycardia pacing and multiple others by 32 J shocks. Transthoracic echocardiogram was notable for moderately-to-severely elevated pulmonary artery systolic pressure estimated at 61 mmHg. Diagnostic angiography demonstrated no significant obstructive disease of the left coronary system. However, the right coronary artery (RCA) appeared to originate anomalously from the left coronary cusp (Supplementary Material Video 1) and course anteriorly with features concerning for a malignant course. Coronary computed tomography angiography confirmed the interarterial course with slit-like compression of the ostial and proximal RCA (Figure 1).
Cardiothoracic surgery was considered for an unroofing procedure; however, the patient was determined to be a poor surgical candidate due to his severe pulmonary comorbidities. The multidisciplinary decision was made to proceed with a percutaneous coronary intervention (PCI) of the anomalous RCA with intravascular ultrasound guidance, which showed compression of the proximal portion of the vessel. He underwent successful placement of a XIENCE Sierra 4 mm × 22 mm drug-eluting stent (Abbott Vascular; Supplementary Material Video 2).
Our patient was closely monitored in the cardiology clinic and at 3-month follow-up, and remained asymptomatic without evidence of recurrent ventricular arrhythmia on remote monitoring of his ICD.
Discussion
AACOA from the inappropriate sinus of Valsalva is a rare congenital anomaly. Our case represents an instance in which the RCA originated inappropriately from the left coronary sinus (i.e. anomalous right coronary artery [ARCA]), and coursed interarterially between the aorta and pulmonary artery. Multiple subtypes exist and there is variation in how cases are classified. AAOCA may arise from the aorta by a separate ostium, shared or common ostium, or as a branch vessel, and can be further characterised by one of five course subtypes: interarterial, subpulmonic, pre-pulmonic, retroaortic or retrocardiac. While the true prevalence of AAOCA remains unknown, the frequency of interarterial ARCA within the general population is estimated to be around 0.23%; approximately sixfold the prevalence of interarterial anomalous left coronary artery.6
The underlying pathophysiology leading to sudden cardiac death in these patients remains not fully understood. Traditionally, the term malignant was used to define anomalous vessels with an interarterial course between the great arteries. Recently, the intramural course and its length have been recognised as more threatening features.3 However, multiple factors likely contribute to limiting coronary blood flow in these patients: intermittent closure of the ‘slit-like’ ostium, kinking of the acute take-off angled anomalous vessel, dynamic compression of the intramural segment and external compression of the interarterial segment by the great arteries.1,3 Additionally, a high course between the aorta and pulmonary artery (where simultaneous systolic compression of the vessels occurs) has been shown to be a factor aggravating blood flow restriction.2
The origin of our patient’s arrhythmia is unclear, with various mechanisms suspected. The non-ischaemic dilated cardiomyopathy itself is the most obvious culprit. However, an underlying arrhythmogenic substrate, either from his cardiomyopathy or the ARCA, is another possible aetiology.4 One unique proposed mechanism includes hypoxic pulmonary vasoconstriction exacerbated by not using his home oxygen with exertion. In this scenario, critical increases in pulmonary artery pressure could intensify compression of the interarterial segment of the RCA, further restricting coronary blood flow and ultimately resulting in the ventricular arrhythmia.
Currently, coronary computed tomography angiography and magnetic resonance angiography are the only class I-indicated tests used to image AAOCA.5 Coronary CT angiography is the preferred imaging modality in many centres due to its rapid scan time, high spatial resolution and lower cost in comparison to magnetic resonance angiography.7,8 In contrast, magnetic resonance angiography provides coronary artery and functional imaging without radiation or iodinated contrast, but incurs lower spatial resolution, increased scan times and higher cost. Invasive coronary angiography holds a class IIa indication for AAOCA, but intravascular ultrasound guidance improves spatial and temporal resolution, and allows for dynamic imaging throughout the cardiac cycle.5
The American College of Cardiology and American Heart Association 2008 guidelines for the management of adults with congenital heart disease recommend surgical repair for ARCA patients with a “course between the great arteries or in an intramural fashion” in the presence of documented ischaemia.5 In these patients, coronary unroofing is the primary surgical approach. Coronary artery bypass grafting is largely avoided, given the concern for graft failure due to competitive flow from the native vessel.9,10
Limited evidence exists to support the routine use of PCI in patients with interarterial ARCA. Currently, it should only be considered as an alternative to surgery in selected older, high-risk surgical patients. It remains unclear how repeated and prolonged episodes of dynamic compression affect long-term stent patency. While one study demonstrated low periprocedural complications and amelioration of presenting symptoms with intravascular ultrasound-guided PCI at 1 year, high rates of in-stent restenosis and recurrent symptoms were observed over long-term follow-up.11 Additionally, selective catheterisation of the anomalous ostium is often challenging because of its ectopic and tangential origin.
Conclusion
AAOCA with interarterial course is a potential underlying cause of sudden cardiac death. The most threatening features are present in patients with an intramural segment. Invasive and non-invasive imaging modalities are recommended for anatomical assessment. In patients with interarterial ARCA and a history of significant symptoms, unroofing is the established and preferred surgical procedure. However, PCI represents an alternative method in select cases. More data are needed to improve risk stratification in this patient group.