Venoarterial extracorporeal membrane oxygenation (VA-ECMO) causes an increase in left ventricular (LV) afterload, reducing forward blood flow, increasing end-diastolic pressure, increasing pressure–volume work and potentiating LV distention.1 Increased LV afterload is associated with poor clinical outcomes. ECPELLA, or the combined use of ECMO with Impella, a percutaneous microaxial pump, can limit this deleterious effect. However, the optimal method, timing and potential reduction of myocardial injury remains to be defined.
Dr Everett’s research hypothesis is that pre-emptive ECPELLA, or Impella activation before VA-ECMO initiation, significantly limits the ECMO-mediated increase in LV pressure–volume area (PVA) and reduces infarct size compared to bailout ECPELLA, or Impella activation after VA-ECMO initiation. LV pressure–volume loops, pressure areas, arterial pressures and infarct size were assessed in adult swine with 90 minutes of coronary artery occlusion, followed by 30 minutes of pre-emptive ECPELLA.
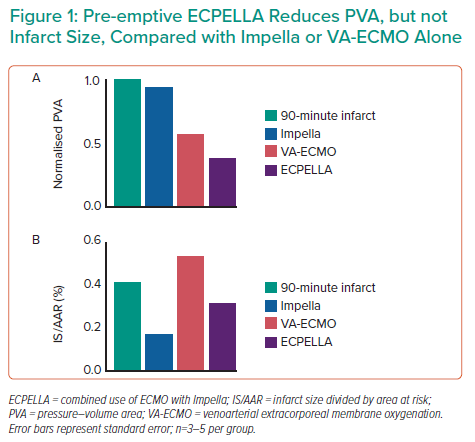
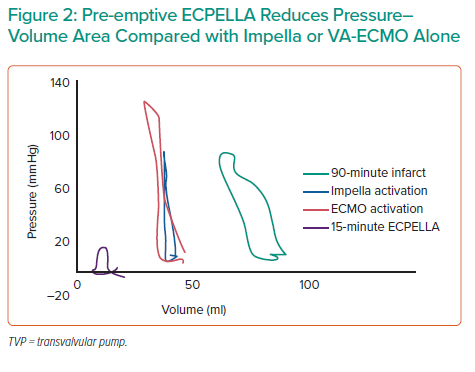
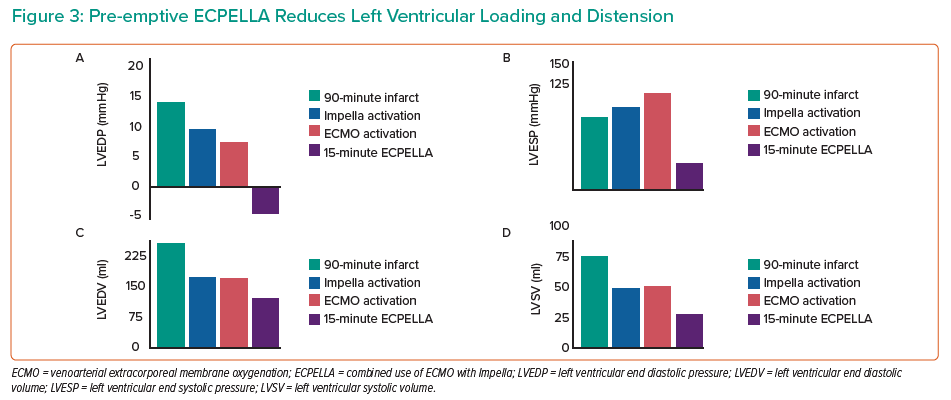
Results from four animals demonstrated that pre-emptive ECPELLA reduces PVA compared with either Impella or VA-ECMO alone (Figure 1). Impella vents the LV, so subsequent VA-ECMO activation increased LV pressure, but not volume, and ECPELLA ultimately reduced overall PVA (Figure 2). Aortic and LV pressure uncoupled after 5 minutes of ECPELLA with maximal PVA reduction observed at 15 minutes. Pre-emptive ECPELLA also significantly reduced LV loading and distension as a function of LV end-diastolic pressure and LV end-systolic pressure (Figure 2). Infarct size was also decreased, but less than with Impella alone, suggesting that VA-ECMO introduces a unique insult that combination therapy with ECPELLA cannot overcome.
Taken together, these data indicate that pre-emptive Impella activation before VA-ECMO significantly limits the increase in LV PVA induced by VA-ECMO compared with VA-ECMO alone. Future directions include the pre-emptive Impella Unloading is Superior to Bailout Impella after VA-ECMO for ECPELLA (PRIMUS) preclinical study to compare pre-emptive ECPELLA to bailout ECPELLA, as well as active versus passive venting in VA-ECMO. LV pressure–volume loops, pressure areas, arterial pressures, infarct size and molecular mechanisms of myocardial injury will be assessed. A clinical trial comparing the effectiveness of pre-emptive ECPELLA to bailout ECPELLA in improving myocardial recovery in shock and acute MI is the ultimate goal of this project.
Dr Everett’s abstract presentation was awarded an Acute Cardiac Unloading and REcovery Research Grant.