Left main stem (LMS) disease is identified in up to 5 % of diagnostic angiography cases.1 It has major prognostic significance due to the proportion of myocardium at risk. Many years ago, the Coronary Artery Surgery Study (CASS) registry demonstrated the superiority of coronary artery bypass grafting (CABG) over medical therapy with a 5-year mortality reduction from 43 % to 16 % in symptomatic patients.2 Since the advent of coronary angioplasty, interventional cardiologists have sought to assess the role of percutaneous treatment of LMS disease. Advances in stent technology, implantation techniques, ancillary imaging and pharmacotherapy have increasingly made this prospect a reality.
Early Experience
The third patient treated by Andreas Gruntzig back in 1979 had LMS balloon angioplasty. The technical result was satisfactory but the patient unfortunately died suddenly 4 months post-procedure.3 In the 1980s, the limitations of ‘plain old balloon angioplasty’ (POBA) in the treatment of LMS disease became apparent. O’Keefe and colleagues showed a 64 % 3-year mortality rate when LMS disease was treated with balloon angioplasty4 and the practice was almost wholly abandoned. Interest in percutaneous treatment was only revived with the introduction of bare metal stents (BMS) and the advent of newer anti-platelet therapies aimed at reducing in-stent restenosis (ISR) and thrombosis.
In the 1990s, promising LMS stent results were published though most stemmed from single centres. Silvestri et al. reported a 1-year survival of 89 % in a high-risk subgroup of patients,5 while Park et al. reported a 91 % 3-year survival rate in a multicentre study.6 The Unprotected Left Main Trunk Intervention Multi-Center Assessment (ULTIMA) registry evaluated the procedural and clinical outcomes after unprotected LMS percutaneous coronary intervention (PCI) was undertaken in 25 centres.7 This showed an overall 24 % 1-year mortality with a better outcome in low-risk groups and only a 3.4 % mortality rate at 1 year. As drug-eluting stents (DES) entered the interventional arena, several centres began reporting their experience in LMS lesions. A systematic review of 1,278 patients by Biondi-Zoccai et al. in 2008 showed that PCI was associated with a 5.5 % risk of death on average and a major adverse cardiac event (MACE) rate of 10.6 %.8
Comparison of Percutaneous Coronary Intervention with Coronary Artery Bypass Graft
Registries
Evidence from the early registries comparing PCI versus CABG suggested that PCI and CABG had a similar MACE rate.9,10 The two observations that emerged were a higher risk of a peri-procedural cerebrovascular accident (CVA) in the CABG group and a higher incidence of target lesion revascularisation (TLR) in the PCI group. The Revascularization for Unprotected Left Main Coronary Artery Stenosis: Comparison of Percutaneous Coronary Angioplasty Versus Surgical Revascularization (MAIN-COMPARE) registry, with data on a total of 2,240 patients, showed comparable outcomes at 5 years though the need for repeat revascularisation with PCI was again highlighted.11 Given the limitations of registry data, more robust evidence was needed to confirm or refute this observation. This spurred the development of randomised controlled trials (RCTs) to gain further insight into the validity of PCI as a viable therapeutic option.
Data from Randomised Controlled Trials
Four RCTs have specifically looked at the cohort of patients with LMS disease to this day.
The Study of Unprotected Left Main Stenting versus Bypass Surgery (LE MANS) was the first RCT and enrolled 105 patients with significant LMS disease (defined as >50 % stenosis angiographically).12 The primary endpoint was the change in left ventricular ejection fraction (LVEF) at 12 months, while the secondary endpoint was a major adverse cardiac and cerebrovascular event (MACCE) at 30 days and 1 year. Surprisingly, there was a statistically significant improvment in LVEF with patients treated with PCI versus CABG (58 % versus 54 %). PCI was also associated with a lower MACE rate at 30 days (2 % versus 13 %) with a MACE being equivalent at 1 year in the two groups. The study did have a number of limitations, including a small sample size, high use of BMS and a lower than contemporary use of left internal mammary artery (LIMA) grafts.
The Synergy Between PCI With Taxus and Cardiac Surgery (SYNTAX) trial remains to this day the largest RCT to date to compare PCI to CABG in LMS disease.13 The LMS subset consisted of 705 patients randomised to receiving either the first-generation TAXUS (Boston Scientific Corporation, US) DES or CABG. The primary endpoint of MACE at 1 year was comparable with 15.8 % for PCI versus 13.7 % for CABG. However, when the cohorts were further subdivided into categories based on lesion complexity, it became apparent that CABG was more favourable for the more complex lesions. The SYNTAX score was developed to objectively quantify lesion complexity and has been fully described elsewhere.14 Stratification into tertiles of syntax score (0–22, 23–32 and >32) revealed equivalent MACCE in the lower two tertiles, but a clear superiority of CABG in the highest tertile. The 5-year follow-up results were published in 2014.15 The results bear a similar trend to the 1-year follow-up with similar outcomes across the whole cohort (36.9 % PCI versus 31 % CABG; P=0.12) but a statistically greater benefit of CABG in the highest risk group based on lesion complexity.
While the SYNTAX trial remains the best evidence-based RCT on revascularisation strategy, it is important to bear in mind that 45 % of patients were excluded from randomisation at the outset due to the complexity of coronary disease. Out of that group, 85 % went on to have CABG. So the evidence we have is from a pre-selected population and this has to be taken into consideration when evaluating whether PCI is an appropriate therapeutic option.
The Premier of Randomized Comparison of Bypass Surgery versus Angioplasty Using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease (PreCOMBAT) trial was a non-inferiority trial randomising 600 patients with LMS disease to either a first-generation Cypher (Cordis Coporation, US) DES or CABG.16 Once again the primary endpoint was MACCE at 1 year. The trial was designed to test whether PCI was non-inferior and indeed proved the non-inferiority of the percutaneous option at both 1-year and 2-years follow-up.
Boudriot et al. studied a smaller sample group of 201 patients and unlike the previous studies, PCI failed to achieve non-inferiority of MACE at 1 year.17 Excess MACE was driven by target vessel revascularisation. Unusually for this type of comparison, stroke was not included as a clinical endpoint, which may well have contributed to the end result.
The results from the RCTs would support PCI as a reasonable alternative in the treatment of LMS disease but the studies have been hampered by limitations. These include relatively small sample sizes, different clinical endpoints among studies and the use of earlier generation DES. This has made it difficult to precisely define where PCI stands.
Meta-analysis
Athappan et al. published a meta-analysis of first-generation DES versus CABG in 2013.18 The authors included 21 observational studies and three RCTs involving a total of 14,203 patients. The MACE rates did not differ between the PCI and CABG group up to 5 years though as has been previously noted, PCI was associated with a lower rate of stroke and higher rate of target vessel revascularisation (TVR). When the cases were stratified according to the SYNTAX score, the results were consistent with those of the main SYNTAX trial with comparable results for the lower two tertiles but a clear superiority of CABG for the highest group.
Influence of Stent Type/Lesion Location and Intravascular Imaging
Stent Type
If PCI is to be undertaken in LMS disease, it is crucial to minimise ISR and thrombosis. A meta-analysis by Pandya et al. comprising 44 studies, showed that DES were associated with a better outcome than BMS.19 The Intracoronary Stenting and Angiographic Results: Drug- Eluting Stents for Unprotected Coronary Left Main Lesions (ISAR LEFT MAIN) trials have compared different types of DES. The ISAR LEFT MAIN study compared the two first-generation DES (Cypher sirolimus-eluting stent [SES] and Taxus paclitaxel-eluting stent [PES]) and showed similar outcomes with both types of stents.20 The ISAR-LEFT MAIN 2 study compared second-generation zotarolimus-eluting stent (ZES) and everolimus-eluting stent (EES) and once again showed similar clinical and angiographic outcomes at 1 year.21 Thus, the types of DES from a similar generation do not seem to influence outcome.
Lesion Location
In an analysis of the Drug-Eluting Stent For Left Main Coronary Artery Disease (DELTA) registry comparing ostial/mid-shaft lesion versus distal lesions, Naganuma et al. demonstrated a higher rate of TLR with distal lesions.22 This primarily drove the higher MACE rates and while there was a trend towards higher mortality and MI, this was not sustained after propensity score matched analysis.
Distal bifurcation lesions present a particularly challenging anatomy to treat. Notwithstanding the increased complexity of such lesions, no technique has been standardised as the technique of choice in LMS bifurcation treatment. In an analysis of the cohort from the ISARLEFT MAIN study, the need for multiple stents was an independent predictor of adverse MACE.23 There seemed to be a better outcome with the use of the Culotte technique as opposed to the T-stent technique with an ISR rate of 21 % and 56 % and a TLR rate of 15 % and 56 %, respectively.
Intravascular Ultrasound
The potential catastrophic consequences of stent thrombosis or restenosis in the context of a treated LMS lesion place an even stronger emphasis on appropriate stent sizing and apposition. The use of intravascular ultrasound (IVUS)-guided PCI in this context has never been formally investigated in an RCT, but data from registries suggest improved outcomes with the use of adjunctive intravascular imaging.
Park et al. compared the use of IVUS-guided treatment of LMS lesions with conventional angiography in the MAIN-COMPARE registry.24 At 3 years, there was a tendency towards improved mortality rates in the former group (6 % versus 13.6 %; P=0.063). This effect was more pronounced in patients who received DES (4.7 versus 16 %; P=0.048).
The Spanish Working Group on Interventional Cardiology (LITRO) study used a pre-specified minimum lumen area (MLA) cut-off of 6 mm2 to determine revascularisation of LMS lesions.25 This was based on Murray’s law assuming an MLA cut-off of 4 mm2 in the left anterior descending (LAD) and the left circumflex (LCX) arteries. Patients with a MLA <6 mm2 underwent revascularisation with over half of the cohort (55.2 %) being treated with CABG while those with an MLA >6 mm2 were managed medically. There was no statistical difference in outcome from the revascularised group versus the medically deferred group (cardiac death-free survival 94.5 % versus 97.7 %, respectively). A cut-off of 4.8 mm2 has been proposed by Kang et al. based on correspondence to a fractional flow reserve (FFR) of 0.8, though this has not yet been clinically validated.26
In an analysis of four Spanish registries, De la Torre et al. found better 3-year outcomes with IVUS-guided revascularisation with a statistically significant reduction in mortality, TLR and MI (88.7 % versus 83.6 %).27 This was even more relevant in a subgroup analysis of patients with distal LMS lesions with better survival rates despite the lower number of patients in that subset.
Guidelines
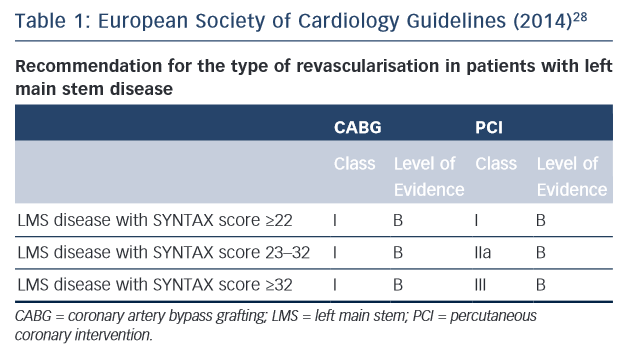
While the results of the two major RCTs are still awaited, the European and American societies have both issued guidelines on revascularisation of patient with LMS disease (see Tables 1 and 2).28,29
CABG maintains a class 1 indication across all anatomical subgroups. It is interesting to note that PCI assumes a stronger position in the European Society of Cardiology (ESC) guidelines with a class 1 recommendation in patients with a low SYNTAX score and 2A for an intermediate score. By contrast, the US guidelines give only a 2A for low scores and a 2B for intermediate scores. Both societies are in agreement about the superiority of CABG for patients with a high SYNTAX score.
On-going Trials
Traditionally, patients with LMS disease as well as additional severe coronary disease elsewhere tend to be referred for CABG assuming decent distal targets are available for grafting. The class 1 recommendation of CABG in high Syntax scores is unlikely to be challenged in RCTs. However, the role in less-severe disease needs to be firmly established. In that respect, we look forward to the results of two trials that hopefully will settle the matter.
EXCEL (NCT01205776) is a multicentre study specifically looking at the treatment of patients with a low or intermediate SYNTAX score (<32). Use of the second generation Xience (Abbott Vascular, US) DES will make the results more relevant to current practice. The primary endpoint is the composite of death, MI and stroke at a mean of 3-year follow-up. It is worth noting that TVR is not included in the primary endpoint but is included as a secondary endpoint. While the original sample size was intended to be around 3,000 patients, recruitment was stopped early for financial reasons. Despite the curtailed sample size, it will still be the largest RCT covering the management of less complex LMS lesions and its findings will certainly influence the management of these patients. The use of IVUS guidance to perform PCI is strongly recommended both pre- and post-treatment.
The Nordic-Baltic-British Left Main Revascularization Study (NOBLE) trial (NCT01496651) is also a multicentre trial comparing the contemporary Biomatrix (Biosensors International Group, Singapore) stent versus CABG. Though the SYNTAX score is not being used for patient selection, the inclusion criteria will yield a population not dissimilar to that of Evaluation of XIENCE PRIME Everolimus Eluting Stent System or XIENCE V or XIENCE Xpedition or XIENCE PRO Versus Coronary Artery Bypass Surgery for Effectiveness of Left Main Revascularization (EXCEL) trial. In addition to the LMS lesions, there should be ≤3 additional non-complex lesions elsewhere to qualify for participation. The primary endpoint is MACE at 2 years.
While the overarching aim of the two trials is to compare PCI with CABG in less ‘complex’ anatomy, it is worth pointing out the subtle differences in inclusion criteria between the two trials. In the NOBLE trial, a significant LMS lesion is defined as having either a visually estimated diameter stenosis >50 % or FFR <0.8. In the EXCEL trial, it is defined as having either a visually estimated diameter of >70 % or 50–70 % with a FFR <0.8 or MLA <6 mm2. Results from both trials are expected in 2016.
The Future
Park et al. recently published a temporal analysis of their experience with ULM PCI over the past two decades.30 It illustrates the achievements in percutaneous treatment of coronary disease with progressively better outcomes as stent technology and profiles evolving over the past 2 decades.
There are other factors that may have contributed to the improvement in clinical outcomes. In addition to improvement in stent technology with the advent of not only DES but also better stent platforms with enhanced drug-delivery systems, the use of adjunctive intravascular imaging and increasing evidence on the efficacy of different bifurcation treatment strategies may have all helped in improving angiographic and clinical outcomes in LMS PCI. With the advent of more potent antiplatelet drugs, the incidence of stent thrombosis has improved. It is important to bear in mind that the techniques of CABG have evolved as well. There is now a greater use of off-pump CABG with associated lower complication rates compared with the traditional surgery as well as almost standard use of LIMA grafts with a better patency than vein grafts. All this means that trials to date are already ‘out of date’ in current real world practice.
The heterogeneity of LMS disease poses a particular difficulty in extrapolating results of clinical trials to the ‘real world’. In that sense, adequately powered RCTs are needed to firmly establish the role of PCI in an era where CABG still remains the gold standard for the treatment of LMS lesions.
Conclusions
While the treatment of LMS disease has historically lain in the realm of the surgery, rapid advancements in the field of percutaneous coronary intervention has provided another viable therapeutic option. Both the American and European societal guidelines have endorsed PCI in patients with less-complex coronary disease while CABG still maintains a class I recommendation across all groups. The data to firmly establish the role of percutaneous treatment are currently lacking and the two on-going EXCEL and NOBLE trials will hopefully take us a step further in clarifying the role of PCI in the treatment of LMS disease.