Since the evolution of interventional procedures from the first right heart catheterisation in 1929, the use of catheter-based interventions has grown exponentially due to new technologies and an increase in coronary artery disease prevalence.1 The risk of radiation injury in a cardiac catheterisation laboratory has always been a topic of debate. American College of Cardiology and American Heart Association guidelines in 2005, 2009 and 2010 emphasised the risks of radiation and the importance of protective measures.2 Most cardiac catheterisation laboratories employ best practice policies to reduce the amount of radiation by using the ALARA (‘as low as reasonably achievable’) technique, as radiation hazard continues to be a major concern.3,4 Radiation exposure mainly emerges as scatter from patients’ bodies and emission from the X-ray tube, which is an occupational hazard in cardiac catheterisation laboratories with far-reaching consequences, including cataracts, cardiovascular disease, brain tumours and thyroid cancer.1,2,5–11 The average dose of radiation in cardiac catheterisation laboratories is about 10–15 mSv for many invasive and non-invasive cardiology procedures.12 Additionally, the radiation exposure dose is proportional to the frequency of procedures performed over time, and cumulative exposure can peak at approximately 100 mSv over a period of 5 years.12 This cumulative radiation exposure manifests as a risk of radiation-induced cancer in one in 100 subjects.12 The annual radiation dose to the head experienced by a cardiologist can range from 20 to 30 mSv, and may rise to 60 mSv/year.13,14 This reveals a dose that is 10-fold greater than that sustained by the whole body.13
High radiation exposure has been linked to malignancies among interventional cardiologists.9,12,15–17 Roguin et al. reported brain and neck tumours occurring in 31 physicians exposed to ionising radiation, including 23 interventional cardiologists, two electrophysiologists and six interventional radiologists.12 Direct radiation exposure to the brain is not well discussed in the literature, but a disproportionate number of left-sided brain tumours can be associated with occupational radiation exposure, given that the face and brain are closer to the radiation scatter in the field.12 Given the high risk of developing brain tumours due to radiation exposure among interventional cardiologists, strategies have to be established to address this problem.13,14,18 Although many steps have been explored, lead caps are one of the noteworthy approaches to shielding the head from radiation.14,18–20 There are limited data available in the literature, and most studies are small and have several limitations in the study design.
This article aimed to provide an overview of the use of lead caps in the cardiac catheterisation laboratory and their role as a protective shield against radiation exposure.
Methods
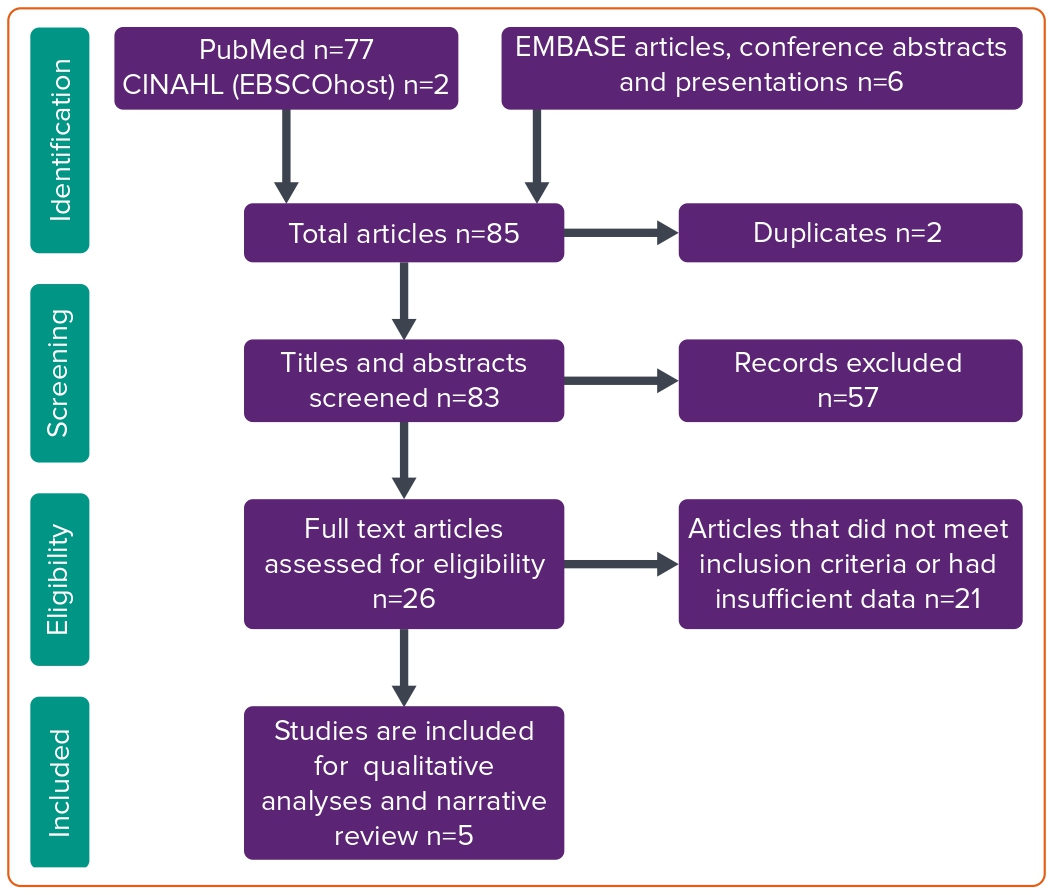
A systematic search strategy was performed in PubMed, Embase (Ovid and Embase Classic) and CINAHL (EBSCO) using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines protocol.21 We used two sets of keywords, such as ‘radiation protection cap’, ‘lead cap’, ‘head protection’, ‘cardiac catheterisation’ ‘cath lab’ and ‘percutaneous coronary intervention’. We used Boolean operators ‘OR’ with similar synonyms, and ‘AND’ between two different keywords. A total of 85 articles were found in online databases, and two duplicates were excluded. Two authors (NJ and MSC) reviewed 83 articles for inclusion of articles to be reviewed that discuss the use of lead caps in the catheterisation laboratory. After this initial review, we selected a total of five studies that were assessed in detail, along with backward snowballing of their references (Figure 1). A full search strategy is mentioned in Supplementary Material.
Results
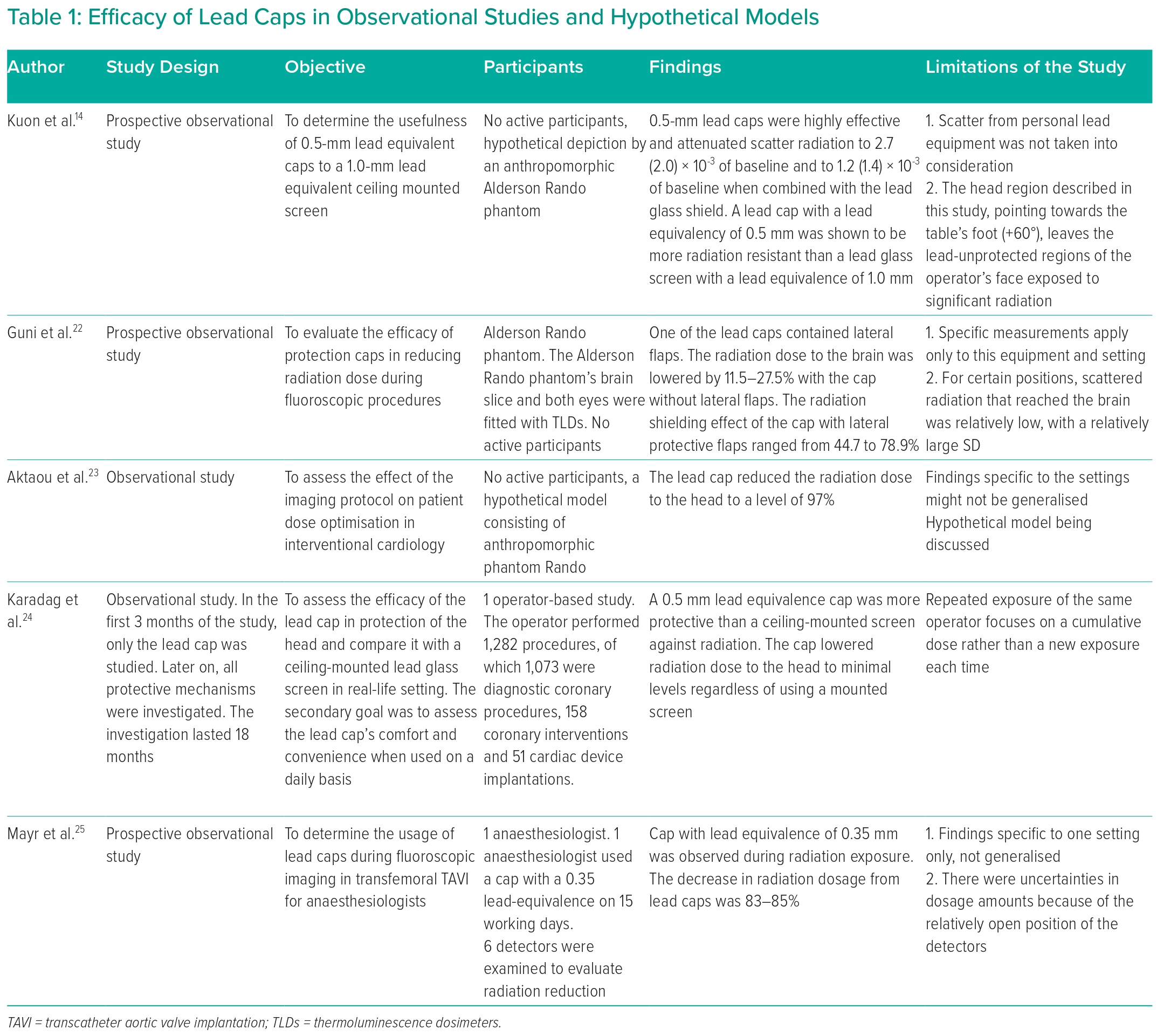
After a careful systematic review qualitative assessment, we included a total of five studies, all of which were observational studies. Three studies had hypothetical stimulation models depicting operators and their positions.14,22,23 In the two remaining studies, each study featured one active participant who was being exposed to radiation at different positions.24,25 The average thickness of the lead caps was 0.27 mm.14,22–25 In one study, the weight of the lead cap used was about 1,140 g.24 The left-sided areas were more commonly exposed to radiation, specifically the left anterior oblique position, left eye and left area of the head.14,22,24,25 All of the five studies documented exposure and subsequent reduction in radiation, as mentioned in Table 1. There was no evidence available for malignancies in the five studies discussed.
Discussion
Effects of Ionising Radiation
Ionising radiation may impact subjects through deterministic and stochastic effects on the human body. Usually, a deterministic effect has a threshold below which the effect does not occur.6,26 Deterministic effects of ionising radiation are meticulously linked to the radiation dose, with dose increments leading to an increase in the severity of the effect.18 For example, interventional cardiologists who do not use radiation protective measures have a dose-dependent increased risk of posterior lens opacities.18,24,26
For stochastic effects, the likelihood of random effects of ionising radiation increases with the level of exposure, but the severity of these effects are not related to the radiation dose.6,26 It appears that there might be no minimum threshold for stochastic effects.6,26 According to one study, chronic exposure increased carotid intima-media thickness and telomere shortening, implying expedited vascular ageing and early atherosclerosis.11
Dose area product measurement and real-time cumulative dosage visualisations are significant advances that have guided operators, especially when treating chronic total occlusion, as these procedures have traditionally been believed to take longer while keeping in mind their complexity.6,14
Radiation Effects on the Head
Several studies postulate the occurrence of brain tumours in interventional cardiologists from radiation exposure in catheterisation laboratories.12,13,17,19 These include glioblastoma multiforme, astrocytomas and meningiomas. The left side of the head appears to be more exposed to radiation than the right in catheterisation laboratories, as revealed in a study that observed a relatively larger number of left-sided brain tumours. This occurrence is possibly linked to radiation exposure. A study in the US examined the risks of the incidence of cancer and mortality among radiation technologists involved in fluoroscopically guided interventional procedures.27 It indicated that technologists who conducted fluoroscopically guided interventional procedures had an almost twofold greater risk of brain cancer mortality as compared with those who never performed similar procedures.27
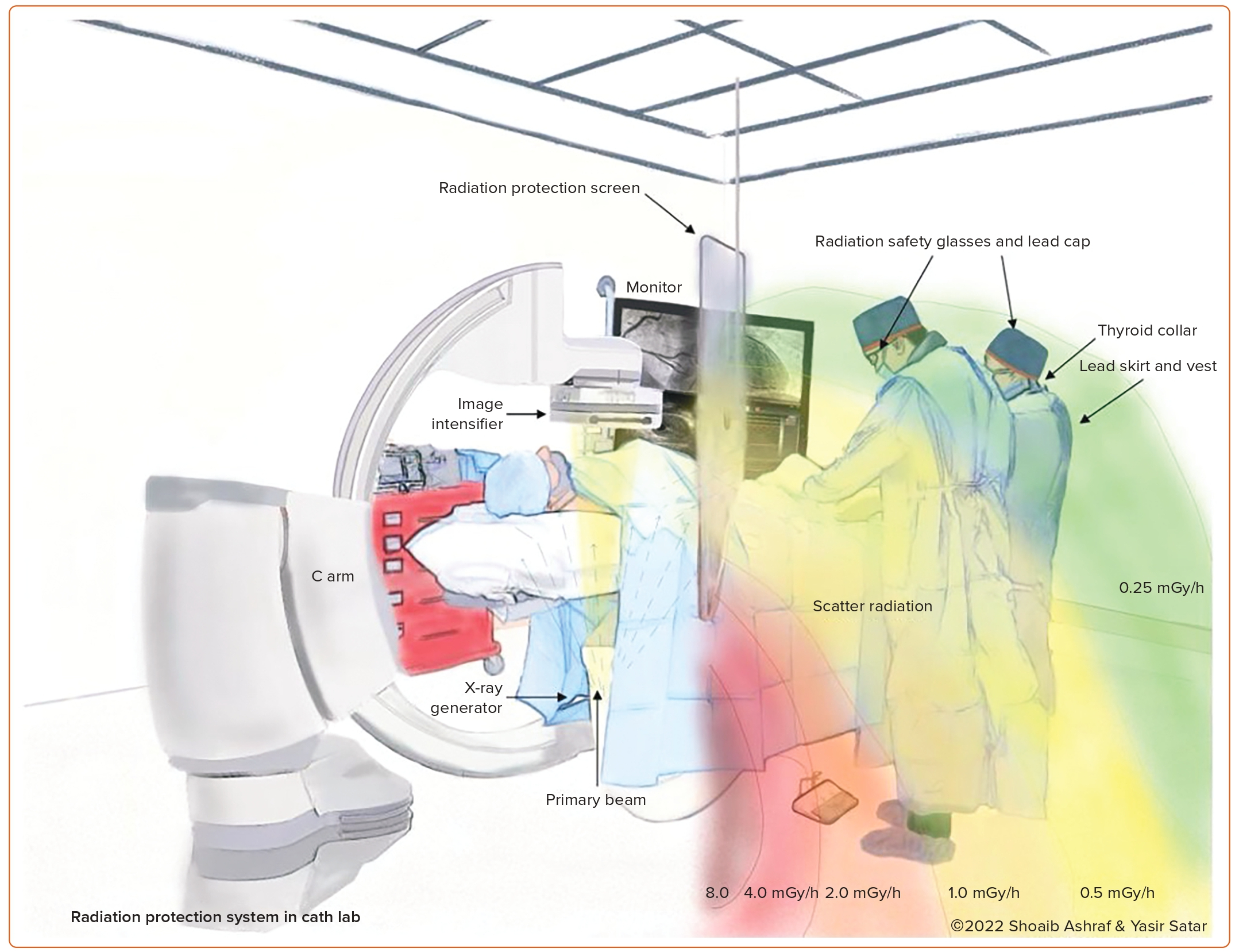
According to a study investigating cognitive impairments after long-term radiation exposure, the exposed cardiologists and nurses had worse scores on visual short-term memory, delayed recall and linguistic abilities than the non-exposed group.28 These findings suggest that head protection is of immense importance for radiation safety among invasive cardiology personnel (Figure 2).28
Radiation Safety
Radiation safety is based on the ALARA principle, as previously mentioned.3,4 The aim of ALARA is to minimise exposure to radiation as much as reasonably achievable while balancing the benefits of medical imaging to achieve a desired clinical or diagnostic result.3,4 According to the International Commission on Radiation Protection, occupational radiation exposure has certain dose limits.29 The International Commission on Radiation Protection advises an effective dose limit of 20 mSv/year averaged over 5 years, and 50 mSv in a single year.29 Furthermore, for the eye lens, an equivalent dose limit of 20 mSv/year, averaged over defined 5-year intervals, with no one year surpassing 50 mSv, is proposed.29 Similarly, 500 mSv/year is the equivalent dose limit for the skin and extremities.29
Methods of Reducing Radiation Exposure
There are multiple methods being used in practice to reduce radiation exposure. Interventional cardiologists are advised to wear dosimeters regularly to monitor radiation dose to observe both radiation exposure and the efficacy of protective equipment.6 Protective equipment includes lead caps, lead gloves, lead glasses, thyroid collars, lead aprons, ceiling mounting glass shields, table lead skirts and patient drapes (Figure 1).7,8,20 Apart from specific protective equipment, a reduction in exposure time and the number of fluorographic images can further limit radiation.6 Lowering the image intensifier to make sure that it is close to the patient and keeping the table as high as possible, including the usage of manifolds, are other measures that will need to be taken routinely to decrease radiation exposure to the staff and the patient. Staff may also use pulsed fluoroscopy with the minimum amount of pulses to obtain images of satisfactory quality and use cine frames only when absolutely necessary.30 Other means of reducing radiation include avoiding steep angles, using collimation when feasible and using technologies, such as stent boost.
Characteristics of Lead Cap
Studies have reported lead caps with several lead equivalencies; for example, 0.25, 0.35 and 0.5 mm.24,25 The average weight of these caps has been reported to be 1.14 kg.8 The cost of one of such cap was reported to be around US$10, but there are limited data on the cost-effectiveness of the equipment.8,18,22
Prevalence of Lead Cap Usage
A study in Germany was conducted to assess compliance to International Commission on Radiation Protection recommendations regarding radiation protection, and included workers from fields of interventional cardiology, radiology and vascular surgery.31 The study included 104 respondents, out of which 54.8% were cardiologists.31 Out of the 104 respondents, only 29.7% reported using caps.31 Of those who wore caps, the proportion of those wearing caps with a lead equivalence of 0.25, 0.35 and 0.5 mm was 43.5%, 39.1% and 17.4%, respectively.31
Efficacy of Lead Cap
Many articles have discussed the use of caps formed from various materials, including lead. In one such study focusing on lead caps in a catheterization laboratory during radiation exposure from transfemoral transcatheter aortic valve implantation, dosimeters were used to measure radiation inside and outside of the lead caps.25 The dosimeters detected a radiation dose of 0.55 mSv outside the cap and 0.08 mSv inside the cap, respectively.25 The decrease in radiation dosage from lead caps was measured at 83–85%.25
Kuon et al. calculated the scatter to entrance skin depending on operator position and the dose area product to evaluate the efficacy of 0.5-mm lead equivalent caps with a 1.0-mm lead equivalent mounted glass screen.14 Lead caps with 0.5 mm were highly effective and attenuated the scatter to entrance skin to 2.7 (2.0) × 10−3 of baseline and to 1.2 (1.4) × 10−3 of the baseline when studied with a 1.0-mm lead glass shield.14 A 1.0-mm lead glass shield decreased the mean dose area product to 54 (29) nSv/Gy cm2, whereas a 0.5-mm lead cap reduced levels to 1.8 (1.1) nSv/Gy cm2.14 In this investigation, the 0.5-mm lead equivalent cap was shown to be more protective than a 1.0-mm lead equivalent glass screen.14 An important finding from this study was that, despite using a ceiling-mounted lead shield to protect the operator’s face and head from scatter radiation, the operator’s head was still at risk of exposure due to secondary scatter radiation being reflected off the laboratory walls, emphasising the importance of lead caps for this secondary scatter radiation.14
Research in Turkey compared the efficacy of lead caps in shielding the head from radiation with a ceiling-mounted lead glass screen in a real-life environment.24 The study involved using dosimeters positioned outside and inside the cap, and outside the apron at the trunk level.24 Regardless of using a ceiling-mounted lead screen, the radiation dosage reported inside the lead cap was <0.1 mSv.24 The study revealed that a 0.5-mm lead equivalence cap was more protective against radiation than a ceiling-mounted screen.24 Moreover, regardless of whether a mounted screen was used, the cap lowered the radiation exposure to the head to minuscule levels.24 When used in routine practice, the lead cap was considered to be comfortable in usage as well.24
Guni et al. observed radiation protection caps in effectively reducing radiation dosage to the brain and eye lens during fluoroscopic procedures.22 Two types of caps with a 0.25-mm lead equivalency were used.22 One of the caps had lateral flaps, whereas the other did not.22 Similarly, caps were affixed to an anthropomorphic Alderson Rando phantom’s head.22 The phantom was placed next to an angiographic table, similar to a first operator’s location during an interventional procedure.22 The Alderson Rando phantom’s brain slice and both eyes were fitted with thermoluminescence dosimeters.22 The cap without lateral flaps decreased the radiation dosage to the brain by 11.5–27.5%.22 Similarly, the cap with lateral protective flaps had a radiation shielding effect ranging from 44.7% to 78.9%.22 According to the findings of this study, radiation protective caps effectively reduce radiation exposure to the brain and eyes.22
Aktaou et al. examined the usefulness of radioprotective devices, drapes and 0.5-mm lead capturing interventional cardiology procedures in a study.23 The incidences of X-ray beams used during interventional cardiology procedures were replicated using pulsed fluoroscopy and high beam energies.23 According to the study, the radioprotective drape, RADPAD Orange, lowered radiation exposure by approximately 50% to the eye and 43% to the whole body.23 The lead cap, according to this study, lowered the radiation dose to the operator’s head until −97%.23 Table 1 summarises the studies discussing the efficacy of lead caps used in respective settings.
Disadvantages of Lead Cap
Lead caps may be uncomfortable to wear due to their weight, which can contribute to neck and back pain.8,32 There is evidence that wearing lead-based protective equipment for long periods leads to lower back and neck pain in interventional radiologists and cardiologists.33 A lead cap may weigh around 1,140 g, which may manifest as orthopaedic problems in operators.7
A study revealed that while metallic lead can be used as a radiation shield, this may impose danger to health.34 Consequently, non-leaded caps were developed to reduce radiation exposure. These include XPF caps lined with barium sulphate and bismuth oxide.35 These caps are as comfortable as normal fabric-based caps with the added benefit of providing considerable protection against radiation.35 A similar impact has been reported for the RADPAD surgical cap.36
In recent times, the advent of new leadless systems developed by RAMPART IC, which has independent adjustable panels, could make a significant difference as they provide comprehensive protection for the whole body.37 Corpath 200 CORINDUS robotic PCI was evaluated in the PRECISE study. Although robotic PCI reduces the radiation to the interventional cardiologist by 95%, an assistant who remains at the patient’s table side has similar radiation exposure to manual operators.38 More data are needed regarding the efficacy of these newer systems. Until then, tools, such as lead caps, need to be strongly considered and employed in day-to-day practice.
Limitations
Study limitations include small sample sizes in most of the included studies and wide variability in the protocols followed in these studies. There is a lack of randomised controlled trials or registries on lead cap usage in cardiac catheterisation laboratories.
Conclusion
Radiation exposure in cardiac catheterisation laboratories is an emerging occupational hazard. While radiation safety has been established using various methods, there is a need to further investigate innovative solutions to reduce exposure to the brain and associated structures. Lead caps are one of the potential solutions to the problem. They come in various styles and sizes, and appear promising by offering the most beneficial protection from radiation exposure to the head. Although results from various studies on lead caps encourage their use, further clinical trials should be conducted to demonstrate the advantages of lead caps usage and establish a safety profile. 
Click here to view Supplementary Material.
Clinical Perspective
- Head and neck malignancies are a concern for cardiologists when it comes to the level of radiation exposure.
- Lead caps have better efficacy in protecting the head against radiation than ceiling-mounted lead glass screens.
- Based on this systematic review, lead caps should be strongly considered in the catheterisation laboratory as part of the personal protective equipment.











Comments