Case–control studies have shown an association between patent foramen ovale (PFO) and cryptogenic stroke, which was observed particularly in patients younger than 55 years without conventional risk factors. The role of PFO closure in preventing stroke recurrence had been uncertain for many years, until four randomised clinical trials in 2017 and 2018 unequivocally demonstrated a benefit, with the number needed to treat between 10 and 46.1–4
COVID-19 has been associated with coagulopathy with increased risk of venous thrombosis, which may, in the presence of PFO, lead to paradoxical embolism and ischaemic stroke.5–9 Given that thrombotic events, including vaccine-induced immune thrombotic thrombocytopenia and formation of myocardial microthrombi, have been documented also after COVID-19 vaccines approved by the European Medicines Agency (EMA), we chose to investigate the occurrence of PFO-associated stroke during the mass COVID-19 vaccination in Slovenia.10,11
Methods
Our prospective observational study was conducted during the mass vaccination against COVID-19 in Slovenia (EU) between 26 December 2020 and 31 March 2022. Consecutive patients with PFO-associated stroke, referred to the International Centre for Cardiovascular Diseases MC Medicor (Izola, Slovenia), were investigated. MC Medicor is a single interventional facility for percutaneous PFO closure in patients older than 18 years in Slovenia, which had 2.107 million inhabitants in 2021.
The likelihood of PFO-associated stroke and the decision for percutaneous closure was always discussed by the PFO team, which included a vascular neurologist, interventional cardiologist and dedicated echocardiographer as previously described.12,13 Patients were eligible for percutaneous closure if they had ischaemic stroke or transient ischaemic attack (TIA) associated with cerebral infarct on CT or MRI, and a PFO with right-to-left shunting at Valsalva documented on transoesophageal echocardiography (TOE). Patients were routinely screened for other possible causes of stroke including large vessel disease, such as aortic or carotid atherosclerosis, hypercoagulable state and paroxysmal AF with 24-hour Holter monitoring if such an event was suspected. If mechanisms other than paradoxical embolisation or lacunar infarct due to intrinsic small-vessel disease were identified, PFO closure was not attempted.
PFO closure, performed under conscious sedation without endotracheal intubation, was guided by fluoroscopy and TOE.12,13 After femoral vein access was obtained, a 6 Fr multipurpose catheter (Cordis, Johnson & Johnson) was advanced over a J-tipped 0.035 inch guidewire (Cook Medical) into the right atrium and through the PFO into the left upper pulmonary vein. After crossing the PFO, a weight-adjusted bolus of unfractioned heparin was given and the J-tipped wire exchanged for a long extra-stiff Amplatz wire (Abbott Vascular). Balloon sizing was performed at the discretion of the operator. PFO was closed by Amplatzer PFO Occluder (Abbott Vascular) using standard procedure. Adequate device shape and position was documented on fluoroscopy and TOE, which was used also for postprocedural evaluation of residual right–left bubble shunting during Valsalva manoeuvre. Following PFO closure, patients received a loading dose of clopidogrel 600 mg and then 75 mg/day for 3–6 months with lifelong aspirin 100 mg/day. Endocarditis prophylaxis was recommended for 6 months after device implantation.
Demographic data and COVID-19 vaccination/COVID-19 disease status were obtained from the medical records and institutional registry approved by the National Ethics Committee (Number 93/11/12). Vaccination data for Slovenia during the study period were collected from the National COVID-19 registry (https//covid-19.sledilnik.org). PFO features were documented on TOE and evaluated by an experienced echocardiographer blinded to the patient’s COVID status. Atrial septal aneurism, floppy septum, Eustachian valve, Chiari’s network and large right-to-left shunt (>20 bubbles during Valsalva manoeuvre) were considered as high-risk features for PFO-associated stroke.14
Continuous variables are reported as mean and standard deviation unless stated otherwise. Unpaired two-tailed t-test was used for comparison between groups. Categorical variables are shown as frequencies and proportions. Fisher’s exact test was used for comparison between groups. p<0.05 was considered statistically significant.
Results
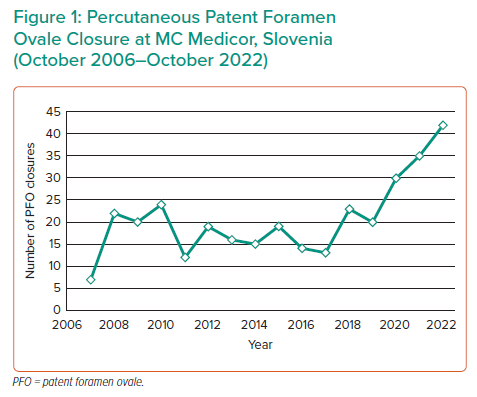
Between 27 October 2006 and 23 October 2022, a total of 323 percutaneous PFO closure procedures were performed at MC Medicor. The annual number ranged from 15 to 25 between 2006 and 2019 and significantly increased to 30 or more in 2020–2022 (Figure 1).
COVID-19 vaccination in Slovenia started on 26 December 2020, and 953,546 inhabitants aged between 18 and 70 years received at least one dose of the EMA-approved COVID-19 vaccine until 31 March 2022.
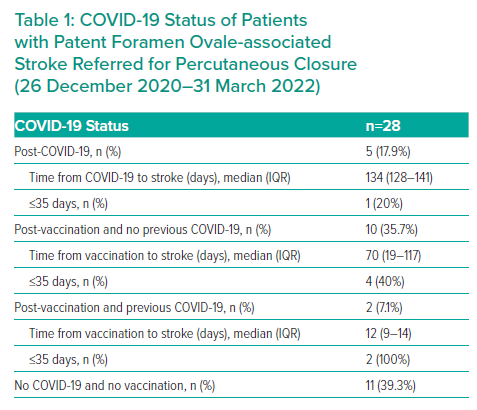
During the study period, 28 patients were referred for PFO closure. Before the index stroke, five patients (17.9%) had COVID-19, 10 patients (35.7%) were vaccinated and two patients (7.1%) were vaccinated following COVID-19 disease, which occurred 299 and 345 days earlier (Table 1). Eleven patients (39.3%) did not have previous COVID-19 and were not vaccinated.
Of the 12 patients with PFO-associated stroke after COVID-19 vaccination, there were nine women and three men aged between 21 and 70 years (Supplementary Material Table 1). Stroke occurred within 35 days after vaccination in six patients (50%). Other predisposing factors favouring deep venous thrombosis included hormone substitution therapy (n=1), contraceptives (n=2) and varicose syndrome (n=1). Clinical presentation of the stroke was heterogenous and included motor dysphasia, paresis, vertigo, ataxia, paraesthesia, headache, diplopia and hemianopia, or a combination of the above. In one patient, stroke occurred after massive pulmonary thromboembolism. Thrombolysis was administered in three patients and none underwent catheter thrombectomy. At hospital discharge, 11 patients (91.6%) had at least one residual ischaemic lesion on MRI.
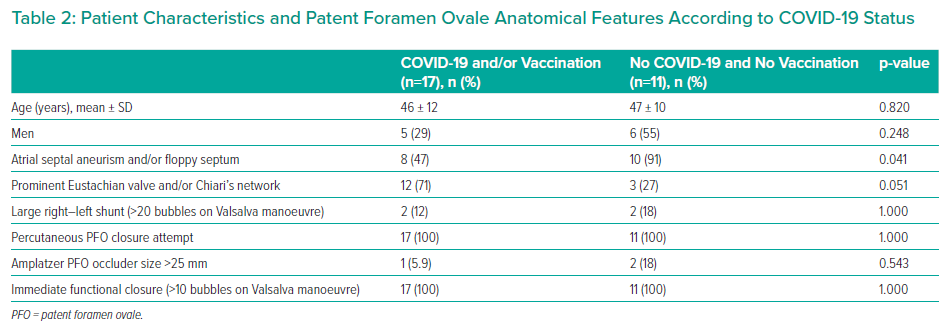
There was no difference in age and sex between patients with stroke after COVID-19 vaccination and/or COVID-19 disease and patients without these events (Table 2). In patients with COVID-19 vaccination and/or disease, atrial septal aneurism and/or floppy septum were present less often (47% versus 91%; p=0.041) and Eustachian valve and/or Chiari’s network more often (71% versus 27%; p=0.051).
Percutaneous PFO closure was successfully performed in all patients regardless of COVID-19 status (Table 2). There was no significant difference in the need for >25 mm closure device and in the rate of immediate postprocedural functional closure between the groups. None of the patients had recurrent stroke or TIA.
Discussion
The proliferation of good-quality observational studies on the potential adverse cardiovascular effects of COVID-19 vaccination has greatly increased our knowledge on myocarditis, pericarditis and, more recently, on arterial hypertension.15 In the present study, we have described a temporal coincidence between COVID-19 vaccination with one of the four EMA-approved vaccines and PFO-associated stroke, with six events (50%) occurring within the 35 days after vaccination. To our knowledge, this potential side-effect of COVID-19 vaccination has not been reported yet.16,17
Although a cause–effect relationship between COVID-19 vaccination and PFO-associated stroke is only hypothetical, systemic inflammatory response and transient increase in spike protein may create a similar prothrombotic condition to that of COVID-19 disease. This may, in turn, facilitate deep venous thrombosis and paradoxical thromboembolism through a PFO, as recently documented in patients with COVID-19 disease.5–9 Interestingly, the number of patients referred to a single Slovenian interventional facility for PFO closure significantly increased during the COVID-19 pandemic by 15–20 cases per year compared with the pre-COVID-19 period. This occurred despite the fact that during the COVID-19 pandemic, priority was given to emergency, urgent and semi-urgent cardiac interventional procedures. This finding may therefore indirectly indicate increased burden of PFO-associated stroke during the time of COVID-19 and mass vaccination. In contrast, the increase in PFO closure procedures in Slovenia might have been related to better and increasing awareness of the benefit of percutaneous PFO closure by our medical community and patients.1–4 In view of this, it will be interesting to observe the future trend in referral of patients with PFO-associated stroke to our interventional facility.
Even if a cause–effect relationship between COVID-19 vaccination and PFO-associated stroke potentially exists, this event seems to be extremely rare and is likely to be identified only during a mass vaccination. Considering that 953,546 Slovenian inhabitants aged between 18 and 70 years received at least one dose of vaccine and given the expected PFO prevalence of 25%, 238,390 persons might have been at risk for PFO-associated stroke. With six strokes occurring close to vaccination (≤35 days), the estimated incidence is only 2.51/100,000. In contrast, given the lack of awareness of this potential adverse event and the preoccupation of the health system with the pandemic, PFO-associated stroke after COVID-19 vaccination might have been underdiagnosed and underreported. Importantly, a similar transient proinflammatory and prothrombotic state is also likely to develop after other vaccines, which, in turn, have never been used in such large numbers in a very short time as were the COVID-19 vaccines. As a result, possible and very rare paradoxical embolism with PFO-associated stroke after vaccination is very unlikely to have been diagnosed and reported earlier.
We further addressed differences in morphological PFO features according to COVID-19 status. Although we noted a lower prevalence of atrial septal aneurism/floppy septum and a higher prevalence of Eustachian valve/Chiari’s network in patients with a history of vaccination and/or COVID-19, these differences do not indicate specific PFO features favouring stroke. It is more likely that they represent the play of chance related to small sample size. We therefore performed routine percutaneous PFO closure regardless of COVID-19 status. Indeed, there was no difference in procedural steps and the effectiveness of closure even when compared with historical controls.13
Limitations
Our study has several important limitations. It is a single-centre observational study with a small number of patients. Although the hospital represents the only national facility for PFO closure in adults, some patients may have not been diagnosed or referred for percutaneous closure due to limited access to the health system during the COVID-19 pandemic. Importantly, a temporal coincidence between COVID-19 vaccination and PFO-associated stroke does not prove a cause–effect relationship, which remains speculative. Indeed, three of the 12 patients were also on hormone therapy/contraceptives, which could provide an alternative explanation for PFO-associated stroke apart from COVID-19 vaccination.
Clinical Perspective
- We have described a temporal coincidence of COVID-19 vaccination with one of the four European Medicines Agency-approved vaccines and patent foramen ovale (PFO)-associated stroke.
- Although our observation does not prove a cause–effect relationship, it should alert the medical community and especially stroke neurologists and interventional cardiologists to be aware of vaccination and/or COVID-19 status in patients presenting with PFO-associated stroke.
- In the case of another mass vaccination against COVID-19 or other disease, larger prospective studies may be warranted.










