Transcatheter percutaneous mitral valve repair (TMVR) using the MitraClip has become a well-established interventional therapy for patients with severe mitral regurgitation (MR). To date, this treatment has been almost exclusively applied in elderly patients rejected for surgical therapy based on the decision of the Heart Team. The basic concept of transcatheter edge-to-edge repair (TEER) arose from the Alfieri stitch double-orifice technique to improve coaptation by approximation of both leaflets.1–3
Until now, EVEREST II is the only study to compare surgical mitral valve repair (MVR) or replacement with percutaneous TEER via the MitraClip. EVEREST II concluded that TMVR showed equivalent clinical outcomes and superior safety compared with surgical MVR. However, conventional surgery was found to be more effective regarding the reduction of MR. Thus, TMVR became the standard interventional procedure for high-surgical-risk patients with severe MR. These patients usually have a combination of being elderly, markedly reduced left ventricular systolic function and multiple comorbidities, rendering them inoperable or high-risk candidates for open-heart surgery.3–5
Worldwide experience in MitraClip therapy has increased over approximately 20 years, with >100,000 patients treated and durable outcomes and proven quality-of-life improvements demonstrated up to 5 years.5 The Carpentier classification describes the mechanism of MR based on mitral leaflet motion and therefore assists in planning mitral valve reconstruction in order to restore normal mitral valve function rather than restoring normal mitral valve anatomy.6 The survival benefit and clinical outcome of mitral valve surgery in patients with severe functional MR and markedly reduced left ventricular (LV) ejection fraction (LVEF) is limited. Recently, mitral valve surgical techniques have relied mainly on surgical MVR rather than replacement of the mitral valve.7,8
The German Transcatheter Mitral Valve Interventions Registry (TRAMI) demonstrated equivalent post-TMVR clinical benefits in older (≥76 years) and younger (<76 years) patient groups and found that advanced age was the most common reason for surgical denial and was a predictor of post-discharge major adverse cardiac and cerebrovascular events (MACCE).9 Post-TMVR complications were confirmed to increase significantly with older age, female sex, increased frailty and higher surgical risk score at baseline in the TRAMI registry.
The aim of this study was to evaluate average 2-year outcomes after MitraClip implantation in patients aged <65 years.
Methods
Patients aged <65 years treated by TMVR were included based on retrospective collection and analysis of data from three German heart centres with at least 2 years of experience in MitraClip implantation procedures and a minimum of 100 TMVRs performed in each centre per year. All patients underwent routine 2D/3D transthoracic and transoesophageal echocardiography prior to the MitraClip intervention in order to determine baseline severity of MR assessed biplane vena contracta width (bpVCW) in two perpendicular views, type of MR; degenerative (primary) MR versus functional (secondary) MR, mechanism of MR based on Carpentier classification, mitral valve morphology and anatomical suitability parameters for MitraClip implantation especially mitral valve area (MVA), baseline mean pressure gradient (MPG) across mitral valve and posterior mitral leaflet length.6,10 Because of the complexity of the mitral valve anatomy, MitraClip may not be suitable for some cases. Clinical characteristics were documented, particularly New York Heart Association (NYHA) class III or IV after guideline-directed medical therapy (GDMT) including CRT if indicated. The combination of the Society of Thoracic Surgeons (STS) score and the European System for Cardiac Operative Risk Evaluation (EuroSCORE), along with a Heart Team meeting discussion, was primarily used to estimate surgical risk preoperatively.11,12 Surgical refusal decisions in spite of the relatively young age of candidates were based on high surgical risk scores at baseline due to markedly reduced LVEF and/or associated comorbidities or malignancies, rather than advanced age in our study candidates.
MitraClip procedures were performed under general anaesthesia in hybrid catheterisation laboratories in all three centres, guided by 2D/3D echocardiography. After grasping the mitral leaflets using the MitraClip and before release of the MitraClip from the delivery system, transmitral MPG <5 mmHg was determined using an optimal atropine test before release of MitraClip. A second MitraClip implantation was carried out only when residual MR was more than Grade II and the MPG across the mitral valve was <5 mmHg.13 Follow-up visits were scheduled at 6, 12, 18, 24, 36, 48 and 60 months. During each visit, worsening of dyspnoea based on NYHA functional class and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) were evaluated. Moreover, 3D transoesophageal echocardiography was used to assess the recurrence of significant MR, defined as MR more than Grade II/IV, stability of the MitraClip in position and tissue bridge formation. Any cardiovascular morbidity in form of decompensated heart failure (HF) requiring hospital admission in addition to cardiovascular and all-cause mortality were recorded. Statistical analysis was performed using Statistical Package for the Social Sciences (Version 26) and based mainly on descriptive statistics and comparing means via paired sample t-test. Quantitative variables were presented as mean ± SD if normally distributed. If not normally distributed, the median was calculated in addition. Qualitative variables were expressed as percentages.
Results
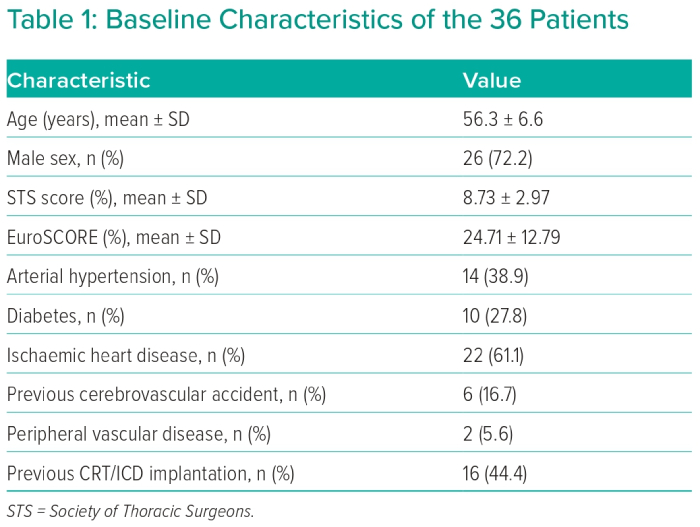
After retrospective collection and analysis of data from three heart centres, 36 patients aged <65 years with severe MR treated by MitraClip were identified. Mean age was 56.3 ± 6.6 years and 26 patients (72.2%) were male. Baseline characteristics of all patients are illustrated in Table 1.
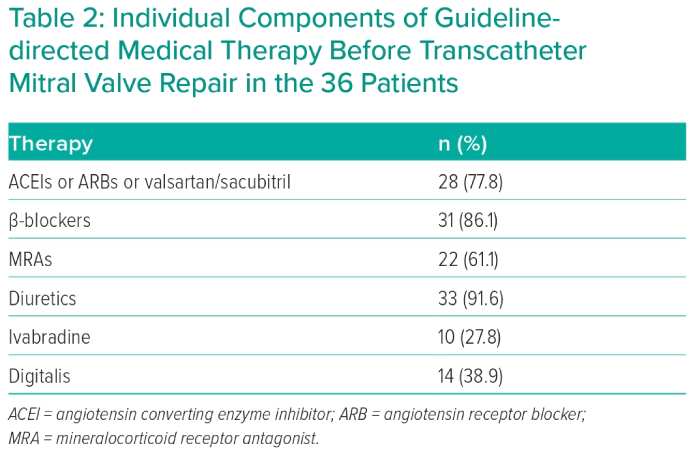
Degenerative MR was detected in 11 patients (30.6%) and functional MR in 25 patients (69.4%). All patients were rejected for surgery by the Heart Team decision due to high surgical risk assessed by STS score (mean 8.73 ± 2.97) and EuroSCORE (mean 24.71 ± 12.79). The Heart Team’s decision relied mainly on severe HF symptoms despite GDMT, including CRT implantation whenever indicated. CRT pacemaker implantation was indicated only in five cases based on QRS morphology and duration, due to markedly dilated LV (LVEDV/BSA 109.8 ± 39.4 ml/m2) and severely depressed LV systolic function (LVEF 27.5 ± 13.6%). GDMT was applied to all study candidates for 3 months with baseline evaluation of NYHA functional class, NT-proBNP in addition to LV dimensions, LV systolic function and MR grade. After that, re-evaluation of the above-mentioned parameters after 3 months of GDMT was performed. GDMT, unless contraindicated in whole or in part, consisted of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers or valsartan/sacubitril, diuretic, mineralocorticoid receptor antagonist in addition to β-blockers with gradual and slow up-titration of β-blockers to the maximum tolerated dose. Individual components of GDMT before TMVR are illustrated in Table 2.
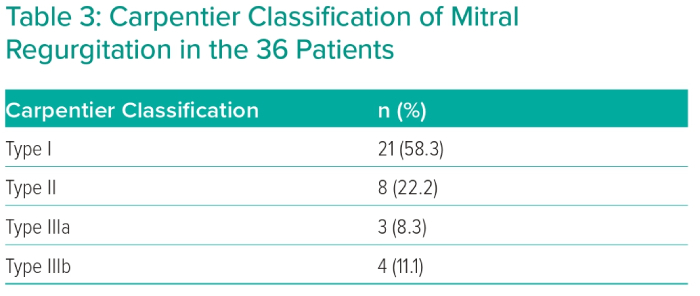
The severity of MR was assessed by calculating the average of bpVCW in two orthogonal views (mean bpVCW 8.35 ± 1.87 mm). Mean preprocedural transmitral MPG was 1.81 ± 0.78 mmHg. Echocardiographic aetiological characteristics of MR based on Carpentier classification are shown in Table 3.
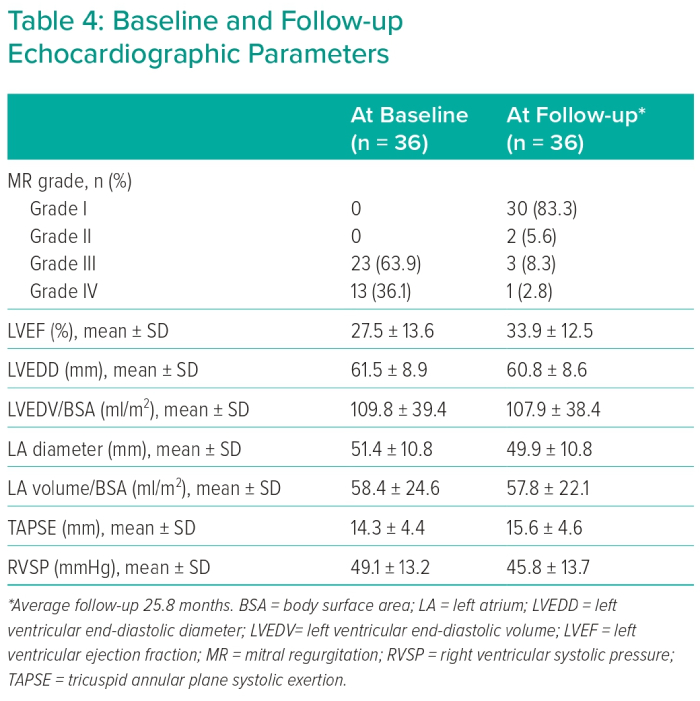
Eccentric regurgitation jets and multiple regurgitation jets were found in 61.1% and 11.1% of patients, respectively. Regarding the aetiological mechanism of MR (Carpentier classification), 21 patients were Type I, showing mitral annular dilatation as the main etiological factor of MR; eight patients were Type II, with leaflet prolapse; three patients were Type IIIa, with mitral leaflet thickening and/or retraction due to fibrosis; and four patients were Type IIIb, with papillary muscle displacement leading to leaflet tethering as a reason for MR. With the help of 3D echocardiographic imaging, cleft mitral leaflet was confirmed in one case and mitral annular calcification was detected in one case. Table 4 shows general echocardiographic parameters.
MitraClip implantation was achieved in all patients, with one, two or three clips implanted in 52.8%, 44.4% and 2.8% of cases, respectively (mean 1.5 ± 0.56 clips per case). Acute procedural success, defined as two or more grades of reduction in MR severity, was accomplished in 88.9% (32 patients). Mean postprocedural MPG was 3.78 ± 1.96 mmHg. There were no procedure-related complications apart from puncture site haematoma in two patients that required surgical evacuation of subcutaneous haematoma and blood transfusion. No surgical repair of the femoral vein was required. Manual compression was sufficient to achieve haemostasis. Early follow-up during the first 30 days after the procedure showed no procedure-related mortality in all centres; however, mortality was recorded in two patients during the first month post-procedure. This was attributed to severe advanced HF in one case and septicaemia after exclusion of infective endocarditis in another case. Over an average of 2 years of follow-up, five other patients died: three because of advanced HF, one because of multi-organ failure and one because of tumour disease. Overall, 47.22% of the study population after TEER experienced at least one HF hospitalisation during approximately 2 years of follow-up. Moreover, eight patients were subject to frequent HF hospitalisation; of these, five died during HF hospitalisation due to intractable HF.
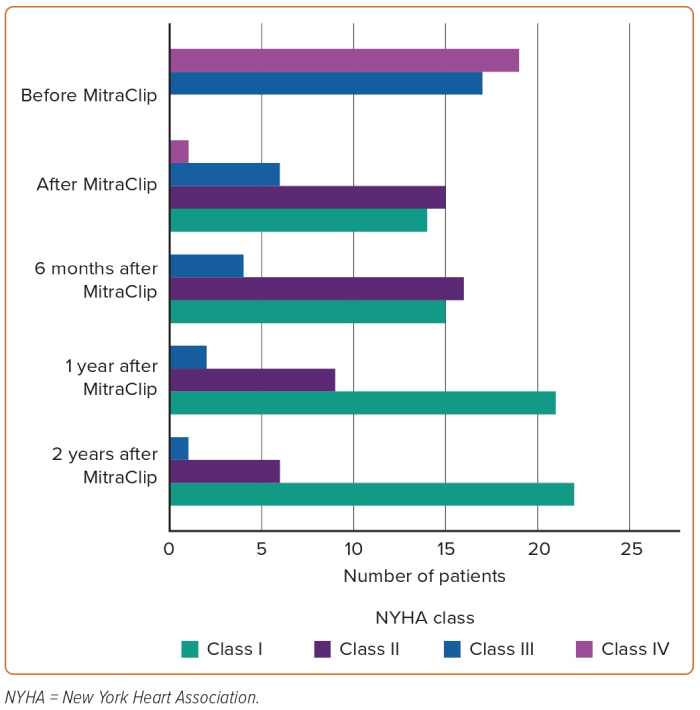
The average follow-up period was 25.8 months and the median was 20 months (25th–75th percentile 12–36 months). A statistically significant difference was detected for NT-proBNP (pg/ml) at baseline (mean 9,870 ± 10,819; median 7,748) compared to follow-up visits (mean 7,645 ± 11,292; median 3,263). Symptomatic improvement during follow-up, defined as NYHA functional class improvement by two or more grades, was achieved in 69% of patients. A second intervention – reclipping – was required in two patients to correct recurrent significant MR. The first case developed recurrent significant MR (Grade IV/IV) 3 days after MitraClip implantation that was accompanied by haemodynamic instability that necessitated the reclipping procedure. The second case had recurrent significant MR (Grade III/IV) associated with worsening dyspnoea and orthopnoea at 6 months regular follow-up and the decision was made to implant a second clip to reduce MR and control symptoms.
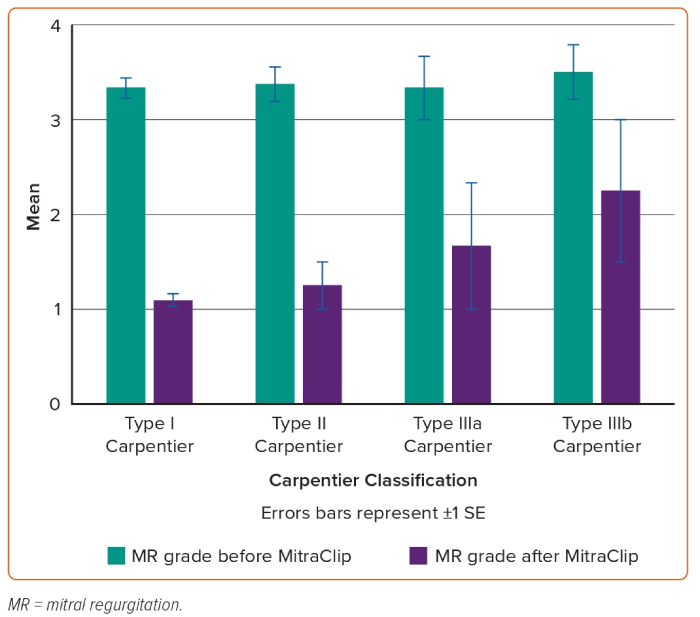
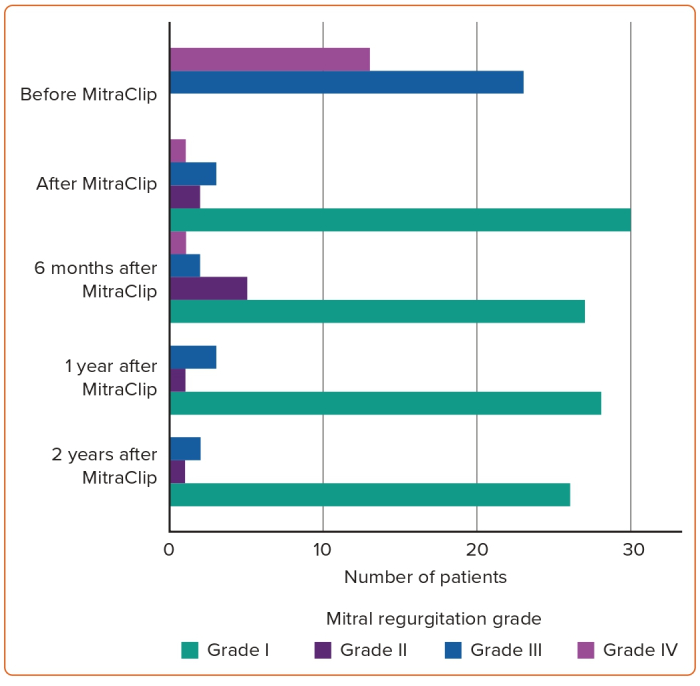
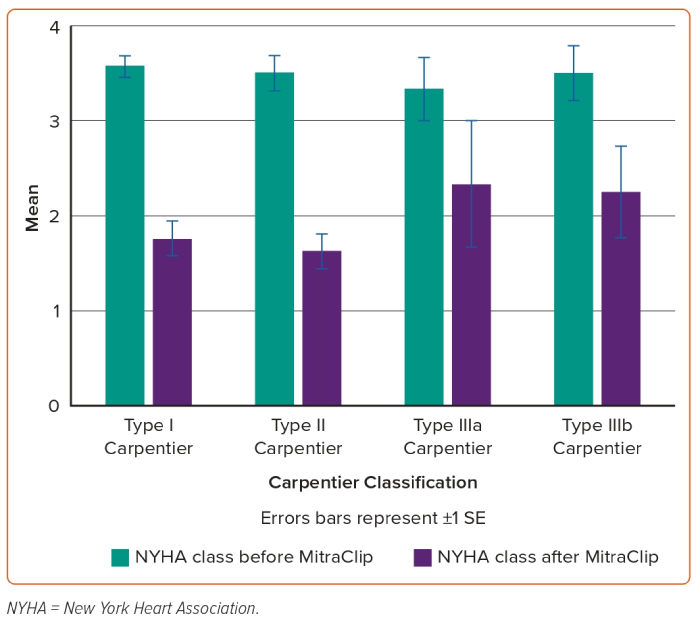
The relation between Carpentier classification and NYHA improvement in addition to persistent MR reduction during clinical follow-up is shown in Figures 1 and 2.
As a bridge to heart transplantation, three patients underwent TMVR to reduce severe MR and subsequently decrease pulmonary hypertension during the cardiac transplantation list waiting period. One patient successfully received cardiac transplantation 3 months after TMVR and is clinically stable throughout 2 years without cardiac decompensation. Another patient received heart transplantation surgery 6 months post-TMVR; after 4 years of regular follow-up after successful heart transplantation surgery, the patient died due to multi-organ failure. The third patient had no further indication for heart transplantation surgery after TMVR because of persistent improvement of HF symptoms during follow-up (Figures 1 and 2).
Discussion
Based on careful review of the literature, to our knowledge, this is the largest study to date dealing with the analysis of post-TMVR clinical outcomes of patients aged <65 years. After at least 2 years of experience in TMVR procedure using MitraClip and at least 100 cases treated every year in each centre, the interventional teams in all three hospitals started the MitraClip procedure in relatively young patients with no surgical options due to markedly depressed LV systolic function, associated comorbidities and malignancy in addition to high STS score and EuroSCORE. These 36 cases represent early experience in the feasibility of MitraClip in relatively young patients, along with 2-year clinical outcomes. The more obvious finding is the persistent clinical response and MR reduction throughout 2 years of follow-up in this category of patients. Based on successful MitraClip implantation in all patients of our study and promising 2-year outcomes such as statistically significant reduction of NT-proBNP, symptomatic improvement and persistent MR reduction, we can state that the procedure is a suitable interventional option for inoperable severe MR patients aged <65 years (Figures 3 and 4).
The same results have been reported in a number of case series and case reports.14,15 Franzen et al. retrospectively studied TMVR via MitraClip therapy in 50 patients (mean age 70 ± 11 years) with end-stage systolic HF and concluded that reduction of functional severe MR in marked LV dysfunction entailed clinical benefit at 6 months of follow-up.16
The 2021 European Society of Cardiology/European Association for Cardio-Thoracic Surgery guidelines for the management of valvular heart disease introduced for the first time the indication for TEER as a class 2b indication for symptomatic degenerative MR patients eligible for MitraClip and inoperable or at high surgical risk.17 Also, guidelines introduced TEER as a class 2a indication for symptomatic functional MR patients, who are inoperable with fulfilment of criteria suggestive of good response to MitraClip and as a class 2b indication for symptomatic functional MR patients, who are inoperable without fulfilment of criteria suggestive of good response.17
The COAPT trial explained that secondary severe MR may be an aetiological factor in the progression of HF with reduced ejection fraction (HFrEF) and that TMVR can change the course of HF disease by reducing the burden of functional severe MR as a risk factor for adverse prognosis.18 However, the MITRA-FR trial failed to detect statistically significant difference regarding symptomatic improvement between GDMT and MitraClip therapy in patients with severe MR and HFrEF.19 This difference is caused primarily by disproportionate MR in COAPT, which can be simply expressed as more severe MR with smaller LV volume and dimensions; this is in contrast to the MITRA-FR population, who had principally proportionate MR, which represents larger LV volume and dimensions in combination with relatively less severe MR.20 Regarding TMVR efficacy, the MITRA-FR population showed less benefit from the MitraClip procedure than the COAPT population due to the difference in the main cardiac pathology of patients included in the trials. In the MITRA-FR trial, advanced LV dysfunction was the primary pathology, whereas severe MR was the primary pathology in the COAPT trial.21
The pre-procedural strategy in this study relied primarily on the implantation of one clip and another clip placement when needed to achieve two or more grades reduction of MR with extreme caution to avoid >5 mmHg MPG across the mitral valve. This was based on Neuss et al. who proved that post-TMVR-induced mitral stenosis with MPG >5 mmHg is a significant predictor of worse long-term outcomes.13 Owing to large bpVCW (mean 8.35 ± 1.87 mm) in these patients, two clips were used in 16 patients (44.4%) and three clips were implanted in a single case (2.8%) with a mean of 1.5 ± 0.56 clip/patient. Recent studies reflect the increasing demand for a multiple-clip strategy. In COAPT, the mean clip number per patient was 1.7 ± 0.7.18 The same pattern was observed in MITRA-FR, whereby 9.4% of patients were treated using three or more clips and 44.9% received two; only 45.7% of patients had one clip implanted.19
In the current study, NT-proBNP levels were found to be significantly reduced during 2 years of follow-up after TMVR. This finding is compatible with Seifert et al., who confirmed that NT-proBNP level decreased significantly in CRT non-responders with severe MR after TMVR.22 Furthermore, in multivariate analysis conducted by Neuss et al., NT-proBNP was found to be a significant predictor of long-term outcome.13 Improvement of NYHA functional class was confirmed post-MitraClip after 4 years of follow-up in EVEREST II, which studied 279 patients who were classified into a TMVR group (mean age 67. 3 ± 12.8 years) and a surgery group (mean age 65.7 ± 12.9 years).23 It should be taken into consideration that all patients of the EVEREST II population were operable, which is not the case in all other MitraClip studies. The final outcome of EVEREST II after 4 years of follow-up stated that the MitraClip population have less severe symptoms, assessed by NYHA class, and better quality of life compared to the surgery population. Nevertheless, the study obviously confirmed that MitraClip was inferior to surgery when it comes to efficacy in reduction of MR, recurrent severe MR, reintervention and mortality. In addition, a remarkable point that we have learned from EVEREST II was that the probability of recurrence of severe MR is low if the initial reduction of MR is maintained 6 months after the procedure.23
This study included three patients who received TMVR as a bridge in order to decrease elevated pulmonary artery pressure before heart transplantation. One of them showed improvement of symptoms and no more indication for transplantation during follow-up and another two patients underwent heart transplantation surgery post-TMVR. The safety of this strategy was established by the International MitraBridge Registry whereby TMVR was used in selected cases as a bridging strategy to cardiac transplantation surgery.24 In addition, 1-year freedom from all-cause mortality, urgent transplantation, implantation of LV assist device and first rehospitalisation for HF was confirmed in 64% of patients.24 Others have reported similar results in case reports and case series.25,26
Our study has some limiting factors that should be considered. These include the retrospective recruitment of cases, the small number of patients and lack of long-term follow-up to clarify the mortality benefit of MitraClip and improvement of HF course after reduction of severe MR. In general, studies with an observational design cannot be relied upon to demonstrate causality.
Conclusion
TMVR in patients aged <65 years not eligible for surgical repair provides satisfactory clinical and echocardiography outcomes at 2 years. Future studies should evaluate the outcomes of MitraClip in this population in a larger cohort. 
Clinical Perspective
- Thirty-six patients aged <65 years were treated via transcatheter mitral valve repair in three heart centres based on surgical denial by the Heart Team due to high operative risks.
- The transcatheter mitral valve repair success rate was approximately 90% with at least two grades of reduction of mitral regurgitation; during the first 30 days, no procedure-related mortality was detected.
- N-terminal prohormone of brain natriuretic peptide levels at baseline were significantly reduced during follow-up visits (p<0.01); New York Heart Association functional class improvement was achieved in 69% of patients.
- Transcatheter mitral valve repair in patients aged <65 years was safe and effective with satisfactory clinical outcomes at 2 years in this cohort.
- In patients aged <65 years at high surgical risk, transcatheter mitral valve repair could be a reasonable option for the management of severe mitral regurgitation.