Out-of-hospital cardiac arrest (OHCA) poses a significant challenge, being a major cause of mortality and ongoing morbidity. In the UK, >30,000 resuscitation attempts occur annually, with the majority recognised as primarily cardiac in origin.1,2 There is international consensus that optimal management involves transport to the nearest heart attack centre (HAC) for prompt investigation and treatment.3,4
Despite advances in treatment, survival following OHCA is low, with a high rate of neurological injury.5 The EuReCa2 study found that of those with an OHCA on whom cardiopulmonary resuscitation (CPR) was started, 36% survive to hospital and 8% survive to discharge.6 Of those admitted to an intensive care unit (ICU) around half will die before discharge, with hypoxic neurological injury as the primary cause of death.5–9 Clinical decision-making in this context is extremely difficult and early accurate prognostication is desirable, given that management of OHCA patients carries a high clinical cost and is a significant emotional burden to family and caregivers.
In OHCA of primary cardiac origin, an urgent invasive management strategy should be considered. Early coronary angiography (CAG) in patients presenting with ST-elevation MI (STEMI) has proven benefit, however there is uncertainty regarding the best management strategy in OHCA patients with non-ST-elevation MI (NSTEMI).10 Despite a high incidence of significant coronary artery disease in these patients, recent randomised controlled trials have demonstrated no benefit to routine early CAG.7,8,11 However, patients without ST-elevation but with significant haemodynamic or electrical instability were excluded from these trials and therefore it remains unclear whether routine early intervention in this group may be beneficial.
Several clinical scoring systems have been developed to assist early prognostication after OHCA: the Out-of-Hospital Cardiac Arrest score, Cardiac Arrest Hospital Prognosis (CAHP) score, Target Temperature Management (TTM) risk score and the MIRACLE2 score.12–15 These can all aid decision-making regarding the appropriateness of medical interventions, as well as supporting family discussions. While the OHCA, CAHP and TTM scores rely on relatively complex nomograms/equations, the MIRACLE2 score is a simple point-based system easily deployed on patient arrival in hospital.
The aim of this study was to externally validate the MIRACLE2 risk score in an independent cohort of OHCA patients.
Methods
Population
This single-centre retrospective cohort study was conducted at the Bristol Royal Infirmary (BRI) between January 2019 and July 2020. The BRI is a tertiary cardiac centre with access to 24-hour emergency CAG, cardiac surgery and specialist intensive care services. All admission records were screened for cardiac arrest ICD-10 codes (I46.0, I46.1, or I46.9). Records were then manually reviewed, with inclusion of all patients aged ≥18 years who presented with an OHCA of presumed cardiac origin and had return of spontaneous circulation (ROSC). Patients who died before hospital arrival, had evidence of a non-cardiac cause of arrest (respiratory arrest, suicide, trauma, drowning, substance overdose), confirmed intra-cerebral haemorrhage, previous significant neurological disability (Cerebral Performance Category [CPC] 3 or 4) or comorbidity leading to life expectancy <6 months were excluded.
Data Collection
Pre-hospital data were obtained from the South Western Ambulance Service Foundation Trust electronic patient care records and initial Emergency Department written records. Zero-flow time was defined as the time from cardiac arrest to commencement of CPR. Low-flow time was defined as the total CPR time, including periods of CPR after re-arrest in the case of multiple ROSC. Baseline blood results were obtained from the hospital’s laboratory database. Twelve-lead ECG, echocardiography and CAG reports were manually extracted from their respective hospital systems and reviewed. An early CAG strategy was defined as within 24 hours of hospital admission.
Scoring Systems
The MIRACLE2 score is composed of seven variables with a potential total of 10 points. Higher scores predict an increasing risk of poor neurological outcome (CPC 3–5). The score components are: unwitnessed cardiac arrest (1 point), non-shockable initial rhythm (1 point), changing rhythms (any two of VF, pulseless electrical activity (PEA) or asystole; 1 point), any adrenaline dose (2 points), no pupil reactivity at ROSC (1 point), initial blood pH <7.20 (1 point) and age category (≤60 years, 0 points; 61–80 years, 1 point; >80 years, 2 points).
As part of the external validation process, we elected to assess the performance of the MIRACLE2 score against previously reported scores, that is, CAHP, OHCA and TTM.
The CAHP score uses a complex nomogram to generate an overall score from the following factors: age, arrest setting, initial rhythm, zero-flow time, low-flow time, initial blood pH and total adrenaline dose.
The OHCA score uses an equation to compute an overall score, with different weights on the factors: initial rhythm, zero-flow time, low-flow time, initial serum creatinine and initial arterial lactate level.
The TTM score is similar to the MIRACLE2 score but uses a more granular system with differing points for 37 subcategories across 10 main factors: age, arrest setting, initial rhythm, zero-flow time, low-flow time, use of adrenaline, pupillary or corneal reflex at ROSC, Glasgow Coma Score (GCS) motor score, initial blood pH and initial arterial partial pressure of CO2.
Endpoints
The primary study endpoint was poor neurological recovery, defined as CPC 3–5 (severe disability to death) at hospital discharge. The secondary endpoint was poor neurological recovery at 6 months.
Ethics Statement
This study falls outside the scope of the UK policy framework for health and social care research and was a service evaluation at University Hospitals Bristol and Weston NHS Foundation Trust. It is a retrospective analysis of routinely collected anonymised data. The study was carried out in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Statistical Analysis
Categorical variables are expressed as absolute numbers (percentage) and were compared using the Fisher’s exact test for 2 × 2 contingency tables or Pearson’s χ2 test for categorical variables of greater dimensions. Normality was assessed using the Shapiro–Wilk test. Variables not normally distributed are expressed as median (lower quartile–upper quartile) and were compared using the Mann–Whitney U-test.
The relationship between predictor variables and the binary primary outcome variable ‘poor neurological recovery’ was evaluated in univariable and multivariable logistic regression models. Standard SPSS multiple imputation methods, using 50 imputations and pooling of data, were used to handle missing predictor variables.
All tests were two-tailed, and p<0.05 was considered significant. The SPSS27.0.1.0 software package was used to conduct the statistical analysis.
Results
Cohort
There were 682 hospital admissions with an associated cardiac arrest ICD-10 code between January 2019 and July 2020. Exclusions totalled 463 patients: 321 records did not relate to a new presentation of OHCA, 40 patients had an obvious non-cardiac cause for arrest, two were dead on arrival, 33 were aged <18 years old, six had a life expectancy of <6 months and five had significant prior neurological disability. An additional 28 patients did not initially present to our centre (transferred in) and 35 had no medical information available. After exclusions, 219 patients were included in the validation cohort (Supplementary Figure 1).
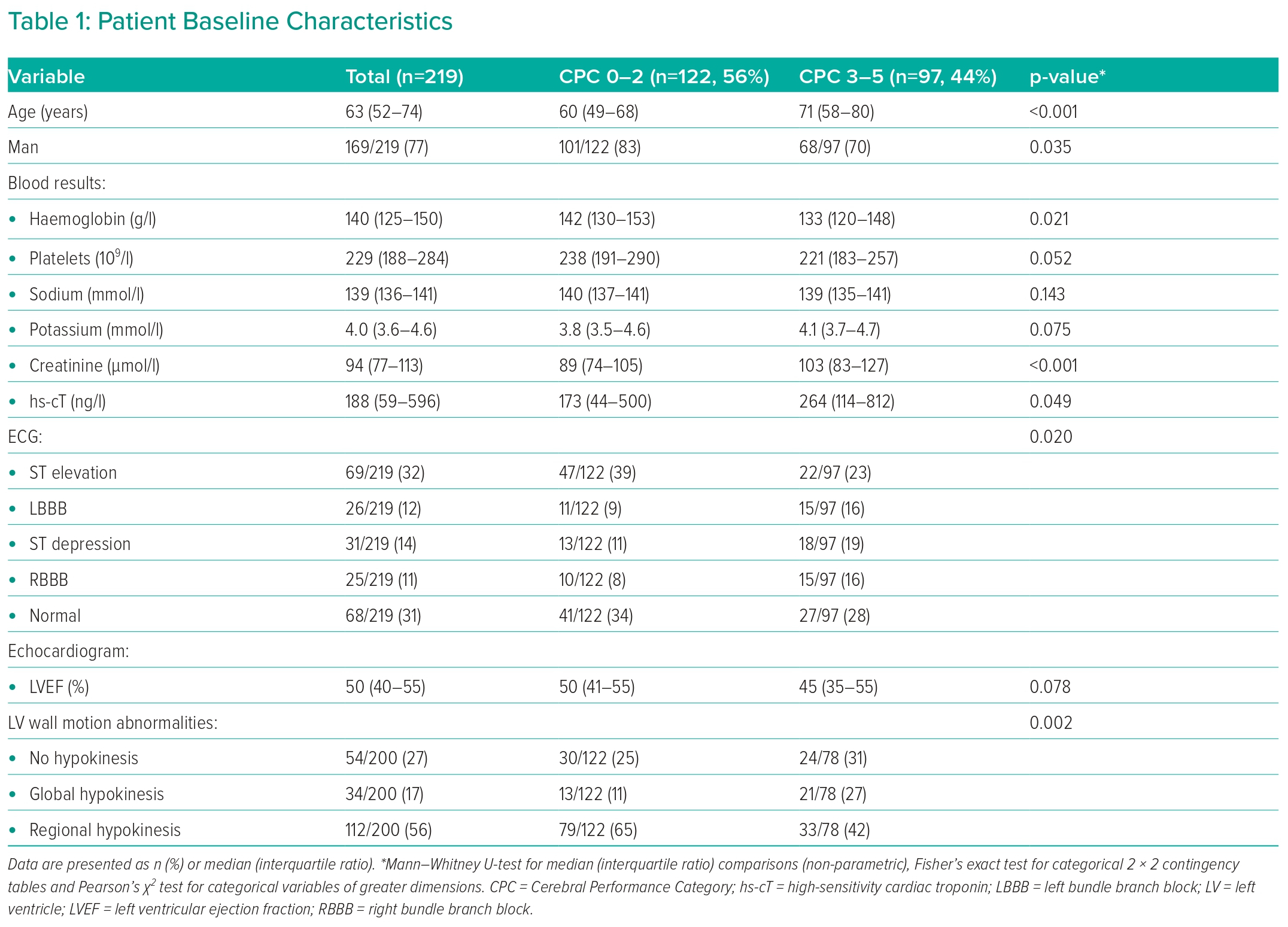
The median patient age was 63 years and 77% were men. Admission ECG demonstrated ST-elevation in 32% and left bundle branch block (LBBB) in 12% of patients. The median left ventricular ejection fraction on echocardiogram was 50% (IQR 40–55%; Table 1).
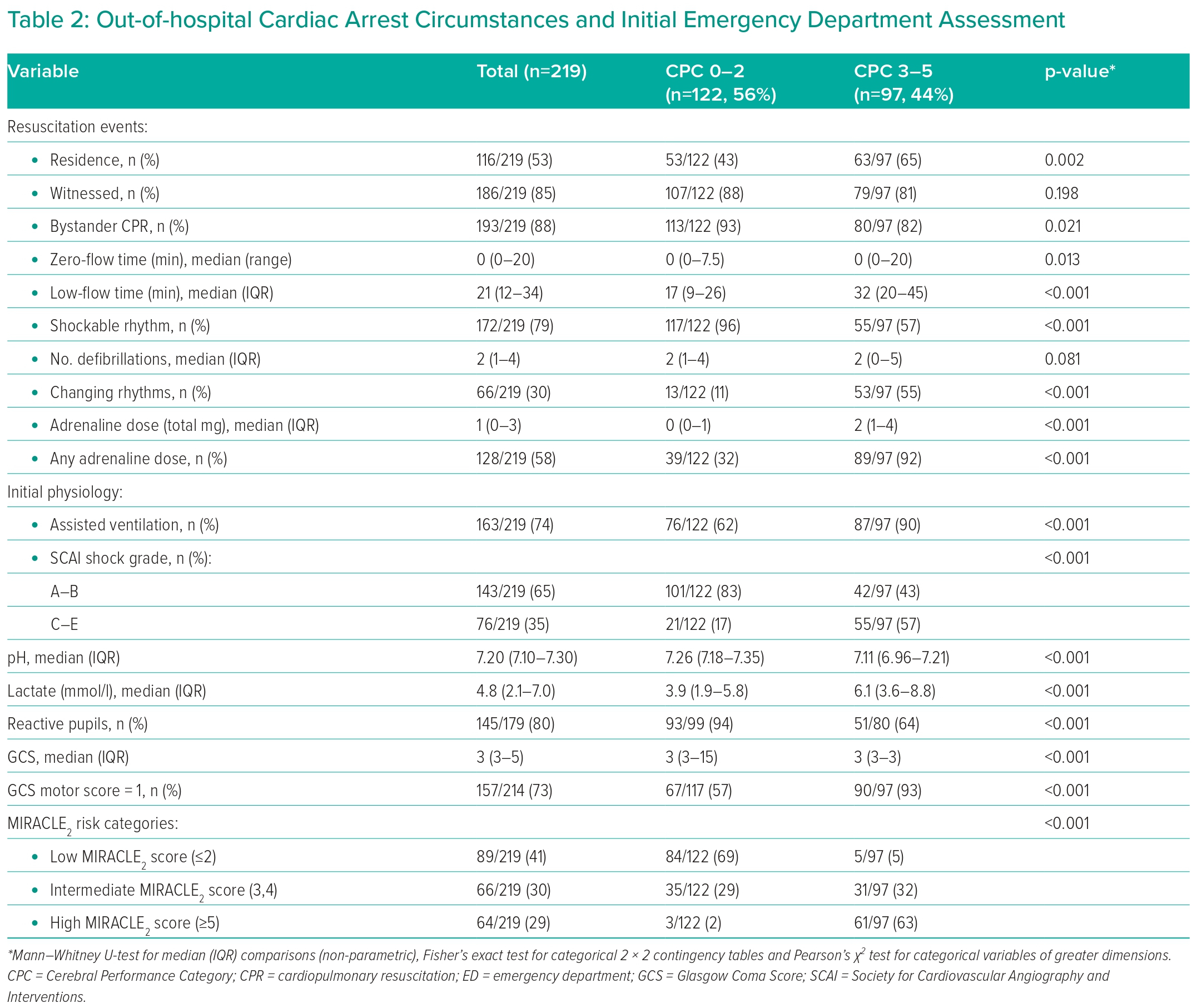
OHCA occurred at home in 53% (116/219 cases) of the cohort, 85% were witnessed and 88% received bystander CPR. The median no-flow time was 0 minutes (range 0–20 minutes) and the median low-flow time was 21 minutes (range 1–139 minutes). An initial shockable rhythm was observed in 79% and a median of 2 defibrillations delivered. Rhythm change during resuscitation was observed in 30%, and the median dose of adrenaline administered was 1 mg. On initial assessment after ROSC, 80% of patients had reactive pupils, the median GCS was 3, and median initial blood pH was 7.20 with a median lactate of 4.8 mmol/l (Table 2).
The majority (80%) of patients were admitted to intensive care, with an equal split crosss MIRACLE2 score risk categories: low (0–2), 33%; intermediate; 34%; and high (≥5), 33%.3,4 Across the same MIRACLE2 risk categories, the 45 patients (20%) not admitted to intensive care were more likely to have a low MIRACLE2 score (71% versus 16% versus 13%, respectively). Significant cardiogenic shock requiring intervention (Society for Cardiovascular Angiography and Interventions [SCAI] shock grade C–E) was observed in 34%.
Overall, good neurological outcome (CPC score 1–2) was observed in 122 patients (56%) at hospital discharge, whereas 97 (44%) had a poor neurological outcome (CPC score 3–5). Outcomes at 6 months were similar and are detailed in Supplementary Figure 1.
MIRACLE2 Scoring and Predictors of Outcome
MIRACLE2 scores were calculated for each patient. Complete data were available in 168 cases (77%) to compute the MIRACLE2 score. Missing data were observed for pupillary response in 40 cases (18%), and initial blood pH for 13 patients (6%). In one patient the initial rhythm was unknown.
The number of patients split across the MIRACLE2 score risk categories: low (≤2), intermediate, and high (≥5), was 89 (41%), 66 (30%) and 64 (29%), respectively.3,4 There was a stepwise increase in the risk of poor neurological outcome with increasing MIRACLE2 score (Figure 1A).
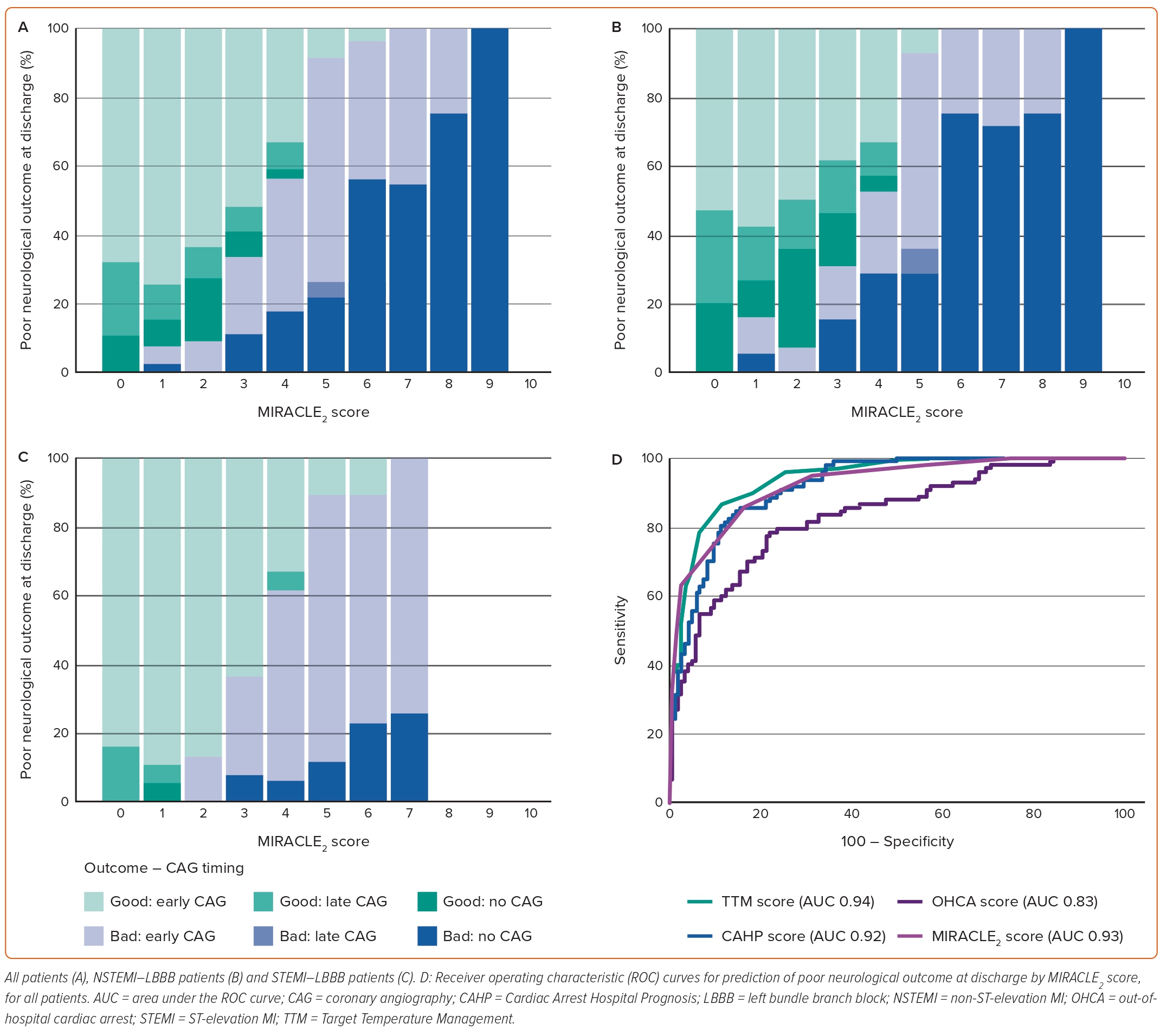
The rates of poor neurological outcome at hospital discharge in patients with a low or high admission MIRACLE2 score were 6 and 95%, respectively. In those with an intermediate score, good or poor neurological outcome was relatively balanced (53 versus 47%, respectively).
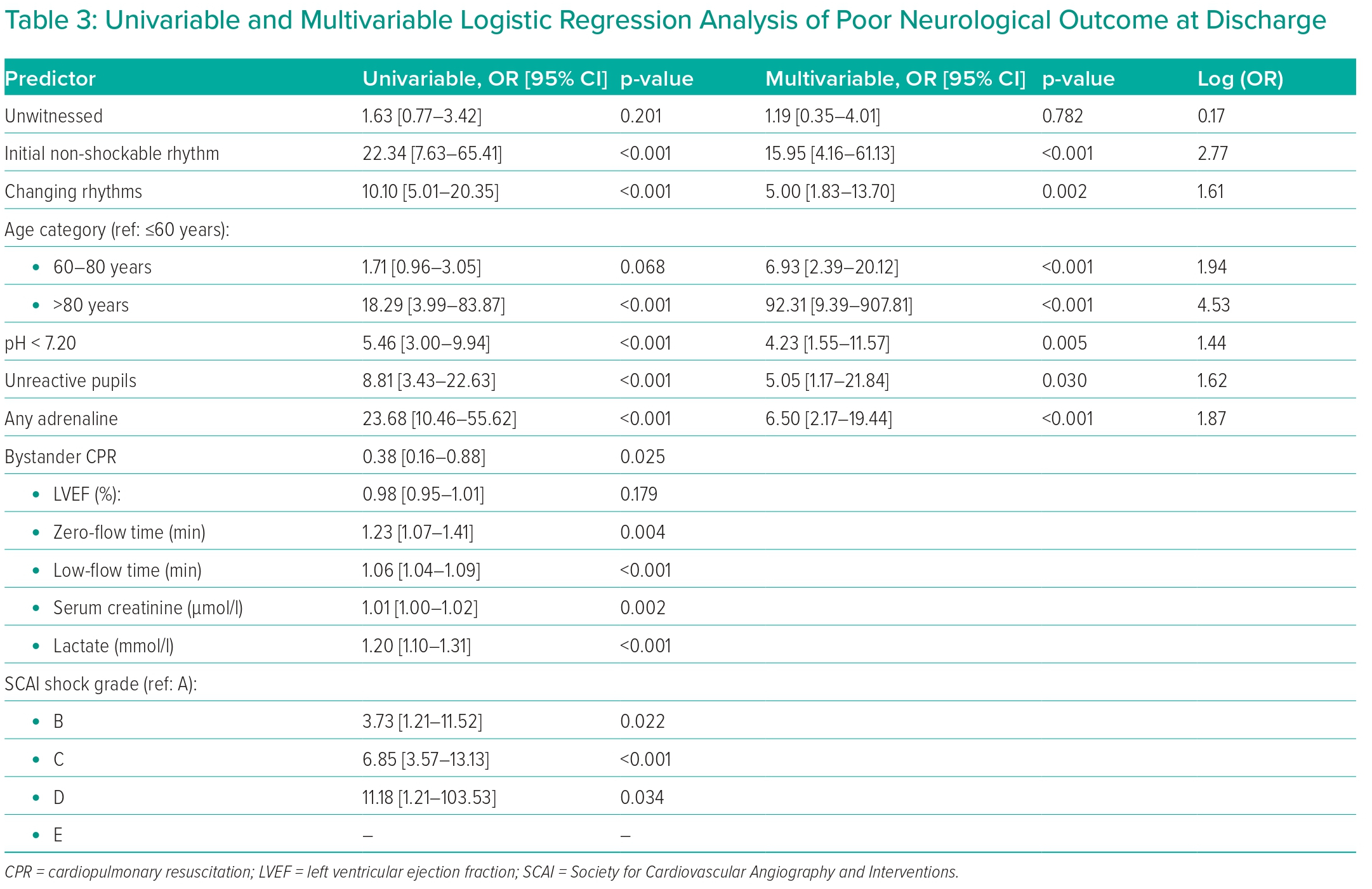
The results of the univariable and multivariable logistic regression analysis are listed in Table 3. The multivariable analysis was conducted incorporating the individual MIRACLE2 score components.
MIRACLE2 Score Performance
Receiver operating characteristic (ROC) curves were derived for the MIRACLE2 score and three other OHCA prognostication scoring systems. When predicting poor neurological outcome at discharge, the area under the ROC curve (AUC) for the MIRACLE2 score was 0.93. The AUC for the CAHP, OHCA and TTM scores was 0.92, 0.83 and 0.94, respectively (Figure 1D). The AUC was almost identical when predicting poor neurological outcome at 6 months: 0.92, 0.92, 0.86 and 0.94, respectively.
The model performance and discrimination characteristics are listed in Supplementary Table 1. A low-risk MIRACLE2 score (≤2) had a negative predictive value of 94% for poor neurological outcome at discharge, whereas a high MIRACLE2 score (≥5) had a positive predictive value of 95%.
Coronary Angiography
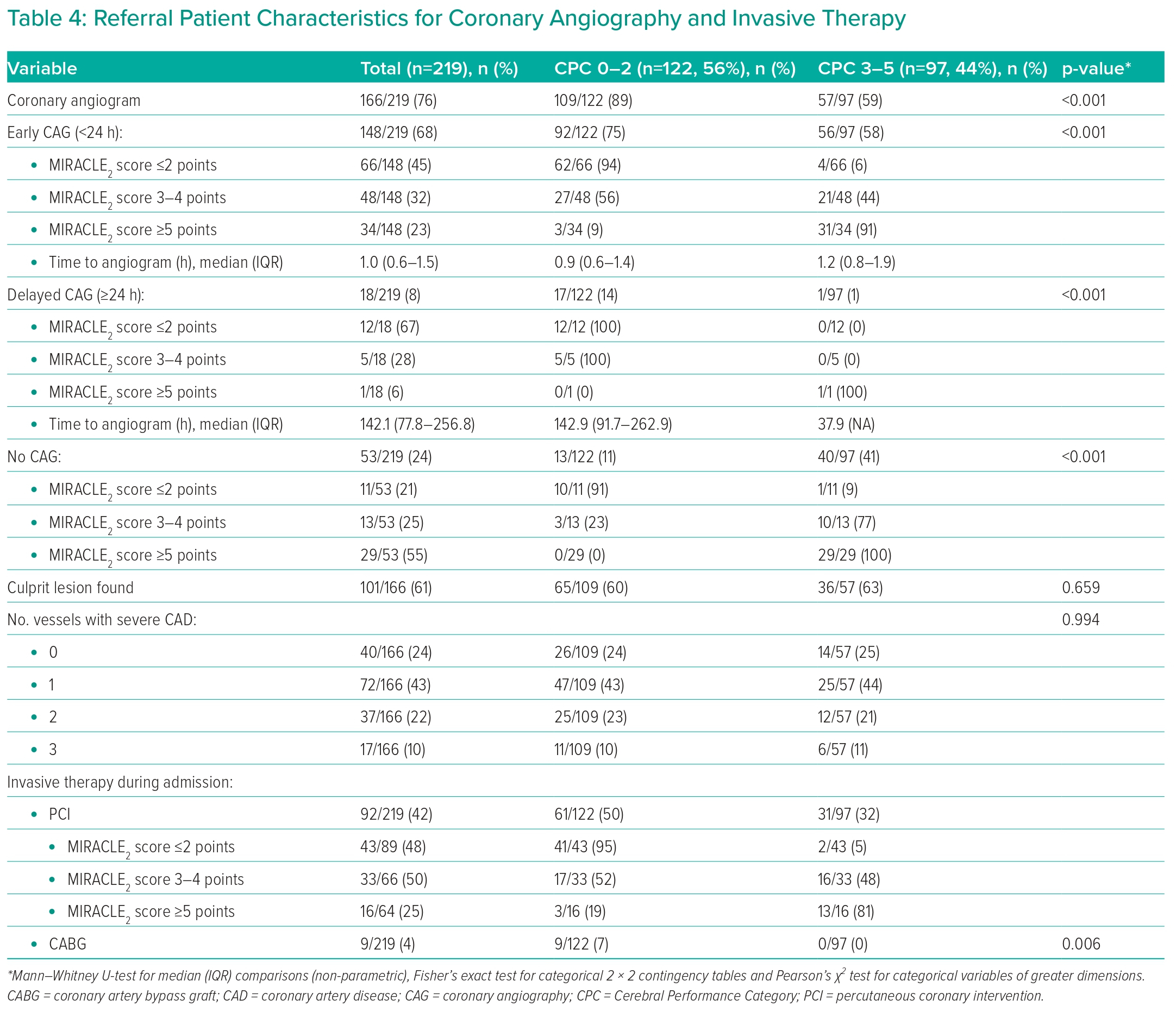
The referral characteristics for patients undergoing CAG and invasive procedures are listed in Table 4. A total of 166 patients (76%) underwent CAG, of whom 148 (89%) underwent CAG ≤24 hours after admission (early-CAG) and 18 (11%), >24 hours (delayed-CAG). The median time to early-CAG was 1.0 hour and to delayed-CAG, 142.1 hours. A culprit lesion was demonstrated in 101 (61%) of the patients taken for CAG, with 92 (55% of laboratory cases, 42% of cohort) proceeding to percutaneous coronary intervention (PCI). Surgical revascularisation was performed in nine cases during admission.
Low MIRACLE2 score patients, compared with intermediate and high score patients, comprised the greatest proportion of those taken for CAG, as either an early-CAG strategy (low 45%; intermediate 32%; high 23%) or delayed-CAG strategy (low 67%; intermediate 28%; high 6%). Conversely, patients with a high MIRACLE2 score were more likely not to undergo any CAG (55%), compared with patients with either intermediate (25%) or low (21%) scores.
In the 148 patients taken for early-CAG, a low MIRACLE2 score was associated with higher rates of good neurological outcome at discharge, compared with intermediate or high scores (97% versus 56% versus 9%, respectively).
In the 18 patients who had a delayed-CAG strategy, all of the low-risk (n=12) and intermediate-risk (n=5) patients had a good neurological outcome at discharge. The one patient with a high MIRACLE2 score had a poor neurological outcome.
In the 53 patients who did not undergo CAG or PCI, a low MIRACLE2 score was associated with a 91% chance of good neurological outcome at discharge. The 77% of patients with an intermediate MIRACLE2 score, and 100% of those with a high MIRACLE2 score, however, had a poor neurological outcome.
The median length of hospital stay was 9.0 days. Good neurological outcome was associated with a significantly longer length of stay compared with those with poor neurological outcome (16.0 days versus 4.0 days, p<0.001).
Discussion
This study provides external validation of the MIRACLE2 score as a simple risk stratification tool, predicting poor neurological outcome following OHCA of presumed cardiac origin. A high-risk MIRACLE2 score ≥5 predicted poor neurological outcome with a specificity of 97.5%. Conversely, a low-risk MIRACLE2 score ≤2 predicted good neurological outcome with a specificity of 94.9%. Intermediate-risk MIRACLE2 scores are associated with less prognostic certainty, with approximately half (32/68, 47%) of patients going on to have a poor outcome.
MIRACLE2 Score
The MIRACLE2 score was developed from a range of demographic and clinical factors.15 Its authors identified a simplified model of seven independent predictors of poor outcome with the highest statistical association, clinical relevance, and practical applicability. The ability of the MIRACLE2 score components to predict neurological outcome in our cohort was assessed using a multivariable logistic regression model and demonstrated good agreement with the original published data.15 The variables associated with the greatest increase in odds of poor neurological outcome were: age category, initial non-shockable rhythm, and any use of adrenaline.
Older age is associated with poorer neurological outcomes after OHCA, reflecting higher comorbidity and lower physiological reserve.16 In our cohort there was a 92-fold increase in the likelihood of poor neurological outcome in patients aged >80 years, and a sevenfold increase in patients aged 60–80 years, compared with those <60 years old. This compares to a 21-fold and threefold greater likelihood in the original MIRACLE2 cohort.15
OHCA patients presenting with ventricular tachycardia or VF have a higher survival rate than those in PEA or asystole.17 Shockable rhythms are more commonly associated with primary cardiac aetiology and more effective treatment strategies. An initial non-shockable rhythm had a 16-fold higher likelihood of poor neurological outcome compared with an initial shockable rhythm. Our results are broadly in agreement with those for the original MIRACLE2 cohort, who had a fivefold increased likelihood of poor neurological outcome.
The role of adrenaline in OHCA remains uncertain, with survival benefit but no neurological benefit observed in randomised trials, although it remains a part of the European resuscitation guidelines.18,19 The use of adrenaline may reflect the complexity of resuscitation, with greater use in those with multiple cycles and prolonged downtime. Regional variation in adrenaline use has been reported, and use of adrenaline in our cohort was lower (58%) than in the original MIRACLE2 cohort (72%).15,20 However, the increased likelihood of poor neurological outcome with any adrenaline use remained comparable (6.50-fold versus 7.67-fold).
Low pH on initial blood gas after OHCA commonly reflects both respiratory and metabolic (lactic) acidosis. We found that pH <7.2 was associated with a fourfold increase in the likelihood of poor neurological outcome, consistent with the original MIRACLE2 cohort (2.26-fold increase).15
Pupillary reflex is a more controversial proxy of neurological insult due to its subjective nature, with a risk of over-reporting reflex absence.21,22 A study of >10,000 post-OHCA patients described lack of pupil reactivity in 64% of cases, but on follow-up 10% of these patients had a favourable recovery.23 However, hypoxic brain injury occurs within minutes of a low-cardiac output, and in the absence of one universal clinical finding signifying futility, pupil reactivity remains a valued predictive parameter.24,25 In our cohort, non-reactive pupils were associated with a fivefold increase in the likelihood of poor neurological outcome, comparable with a 2.49-fold increase in the original MIRACLE2 cohort.15
Unwitnessed arrest was not significant in our model. Patients coded as unwitnessed totalled 33 (15%), of whom 25 (76%) had bystander CPR once found. The median estimated total downtime compared favourably with witnessed arrest (28 minutes versus 23 minutes, p=0.139), suggesting that the unwitnessed no-flow period was short in this group. This will have had the effect of reducing the predictive contribution of the unwitnessed arrest component of the MIRACLE2 score.
MIRACLE2 Score and Coronary Angiography Decision-making
The majority of our data come from the post-COACT era, but before publication of the TOMAHAWK trial.7,8 These landmark clinical trials have been interpreted by many as a strong mandate not to take patients without ST-elevation to the catheterisation laboratory. Although routine CAG may not be an appropriate strategy in the absence of ST-elevation on ECG, the decision is more nuanced than existing trial data allow for and many factors need to be considered, including haemodynamic and electrical instability and overall chances of a good neurological outcome. It is particularly important to emphasise that patients with haemodynamic or electrical instability were excluded from COACT and TOMAHAWK.
It is important to note that our OHCA service, as indicated by the percentage of ventilated patients undergoing PCI, has been an outlier in the UK national dataset for many years.26 This reflects early adoption of a collaborative multidisciplinary pathway, involving emergency physicians, cardiologists and intensivists, with a low threshold for early CAG. Retrospective application of the MIRACLE2 score to our contemporary data demonstrates a selective decision-making process (24% of patients not undergoing CAG) but suggests that prospective application of the score may avoid unnecessary intervention in some of the highest risk individuals.
For patients who underwent CAG within 24 hours of hospital admission, the median admission-to-laboratory time was comparable to that reported in both COACT and TOMAHAWK (1.0 hour versus 2.3 hours and 2.9 hours, respectively).7,8 Our cohort included STEMI and LBBB presentations, which these trials did not. However, the median time to laboratory was still 1.0 hour in the early-CAG group even after the removal of STEMI and LBBB patients. As expected, the primary outcome was observed less in the STEMI–LBBB cohort (39%) but importantly the outcomes in the TOMAHAWK cohort mirrored the study (48% CPC 3–5).8
In our cohort, a low-risk MIRACLE2 score (≤2) was associated with good neurological outcomes regardless of early-CAG (62/66, 94%), late-CAG (12/12, 100%) or no-CAG (10/11, 91%) strategies. This most likely reflects appropriate individualised clinical decision-making.
A high-risk MIRACLE2 score (≥5) was associated with poor neurological outcomes regardless of CAG strategy. Of the 34 patients with high-risk MIRACLE2 scores who underwent early-CAG, only 3 (9%) had a good neurological outcome at hospital, whereas the majority (31, 91%) had a poor outcome. A similar pattern was seen in the presence or absence of ST-elevation with LBBB on initial ECG, suggesting that a high MIRACLE2 score is a useful prognostic tool regardless of presenting ECG, representing damage already done in this unfortunate cohort.
A MIRACLE2 score ≥5 was 97.5% specific for poor neurological outcome in our cohort. Although a single scoring system will never cover all cases, this suggests that the MIRACLE2 score is a useful tool that can aid in discussions between specialist hospital teams and the patient’s family, to minimise futile treatments and enable better palliative care.
Intermediate-risk MIRACLE2 Score
Our data suggest that much of the prognostication in the low and high MIRACLE2 risk categories can be achieved at the hospital front door using basic clinical information. However, intermediate-risk MIRACLE2 patients have the greatest prognostic uncertainty and represent 31% of our cohort. Good neurological recovery was observed in 35 of the 66 (53%) intermediate-risk patients at hospital discharge. This was similar regardless of presenting ECG (STEMI–LBBB, 16/32, 50%; NSTEMI–LBBB, 19/34, 56%).
Limitations
This study has several limitations. The MIRACLE2 score has been created from a retrospective registry of OHCA patients, and as yet it has not undergone prospective validation. The present cohort attended a tertiary centre with immediate access to specialised care and a dedicated cardiac service, similar to the original cohort; therefore, it is unclear how the score would perform in a centre with a less mature service.
The MIRACLE2 score is based upon an immediate static assessment of a critically ill patient. The pupillary response is one factor that may evolve as a patient progresses through their post-resuscitation care. The response captured in the MIRACLE2 score represents an initial assessment at the point of presentation, typically in the first hour. Ongoing pupillary assessment is required as part of a patient’s neuro-prognostication.
Generation of a dynamic model tracking continuously monitored data may provide greater accuracy of outcome and further guide the complex management of this high-risk population.
Intermediate-risk MIRACLE2 patients represent a difficult population in terms of identifying those most likely to achieve a good neurological outcome. The utility of an intermediate score, in isolation, is low. This group requires careful consideration of the most appropriate approach, on a case-by-case basis, within a multidisciplinary framework.
While this retrospective study provided a large and detailed dataset, there were missing variables, particularly those of pupil reactivity and blood pH measurements.
Conclusion
The MIRACLE2 score demonstrates good prognostic performance and is easily applicable to cardiac-origin OHCA presentation at the hospital front door. Prognostic scoring may assist decision-making regarding early angiographic assessment. 
Click here to view Supplementary Material
Clinical Perspective
- Out-of-hospital cardiac arrest (OHCA) is associated with poor clinical outcome, and consensus on the pathway of care is yet to be agreed. Neuro-prognostication scoring may assist in treatment decision-making but external validation of scores is essential before considering prospective utility.
- This study has validated the MIRACLE2 score as a predictor of neurological outcome after OHCA.
- The MIRACLE2 score demonstrates excellent performance and suggests possible utility in guiding treatment decisions in OHCA patients, particularly for those at highest risk of poor outcomes.